Permanent Dialysis Access Made Easy: Trusted Catheter Kits for Long-Term Use
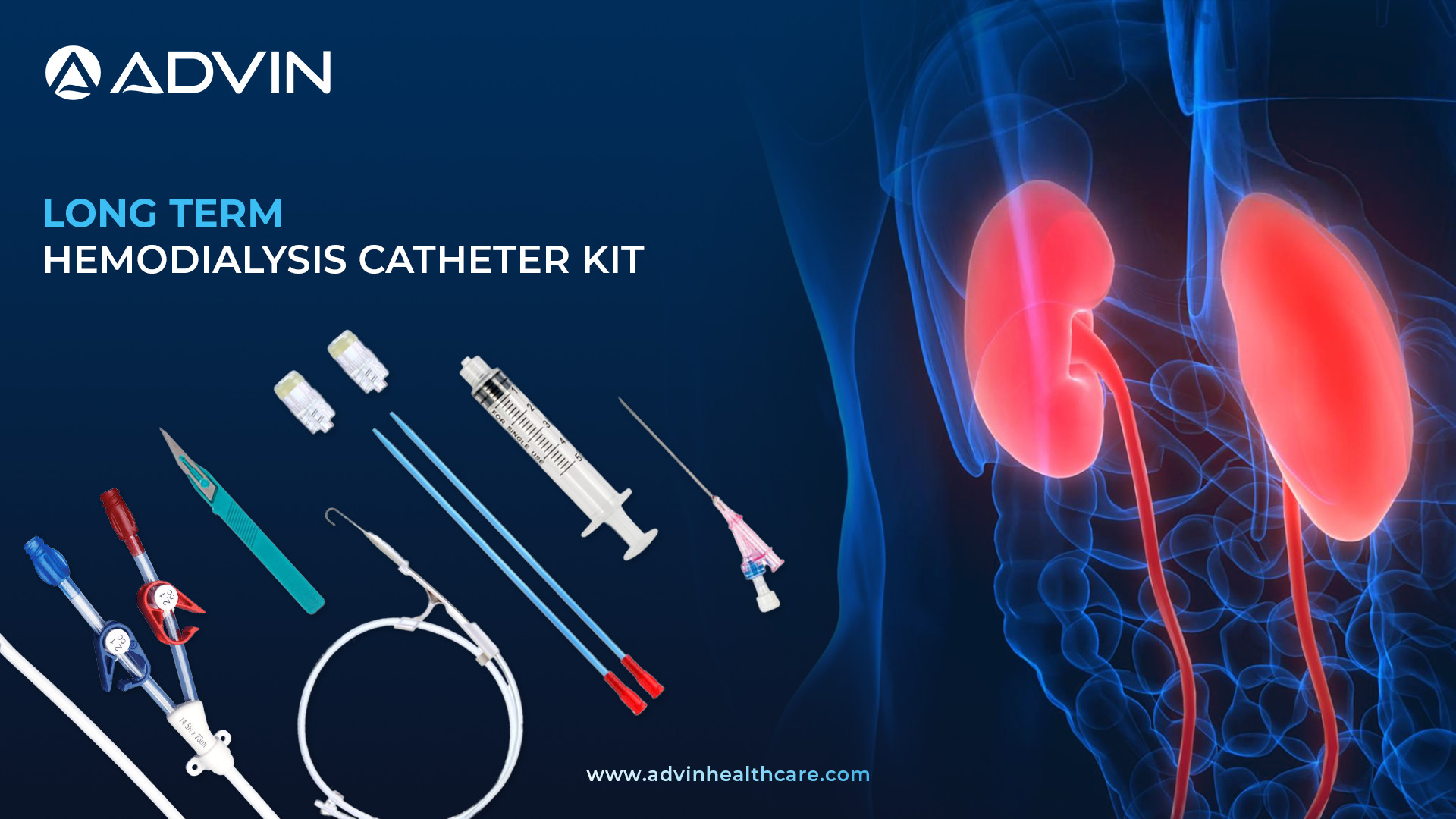
Long-Term (Permanent) Hemodialysis Catheter Kit
The Long Term (Permanent) Hemodialysis Catheter Kit is used to provide vascular access for patients undergoing regular hemodialysis treatment. It is implanted into a large vein, usually in the chest area, and allows repeated access without frequent needle sticks. This catheter is designed for long-term use in patients with chronic kidney disease.
Product History
Permanent hemodialysis catheters have been in use since the late 1980s as a solution for patients who require ongoing dialysis. Earlier, temporary catheters were used, but they posed risks of infection and needed frequent replacement. Medical advancements led to the development of long-term catheters, offering more stability and reduced complications. Over the years, these catheters have become an essential part of chronic renal care across the world.
Long-Term Hemodialysis Catheter Kit – Designed for Comfort, Built for Longevity
Advin Health Care offers a high-quality Long Term Hemodialysis Catheter Kit designed for continuous and reliable vascular access. The kit provides ease of insertion and is made with biocompatible materials that help reduce clotting and infection risks. It includes all necessary components for successful catheter placement in long-term dialysis patients. Advin Health Care ensures safety, comfort, and durability with its expertly engineered catheter kits. The product is trusted by nephrologists and dialysis centers globally.
Surgeries Related to Product
- Chronic Hemodialysis Access Surgery
- Vascular Access Surgery
- Central Venous Catheter Placement
- Tunneled Catheter Insertion Procedure
Instructions for Use
- Ensure the patient is in a sterile environment.
- Identify and disinfect the insertion site, typically the internal jugular vein.
- Insert the catheter using a guidewire and dilator under imaging guidance.
- Tunnel the catheter under the skin to reduce infection risk.
- Secure the catheter and connect it to the dialysis machine as needed.
Top 10 Countries of Surgeries
- United States
- India
- China
- Brazil
- Germany
- United Kingdom
- Japan
- Russia
- Mexico
- South Africa
Advin Long-Term (Permanent) Hemodialysis Catheter Kit Description
Advin Health Care is a trusted manufacturer of Long Term Hemodialysis Catheter Kits, offering reliable and safe vascular access solutions for chronic dialysis patients worldwide.
The Long-Term (Permanent) Hemodialysis Catheter Kit is designed for patients needing ongoing dialysis treatment. It provides secure, long-lasting vascular access using a dual-lumen catheter made from biocompatible materials, typically inserted into a central vein in the neck or chest. The kit includes essential accessories like drapes, syringes, and clamps for a safe procedure. With proper care, the catheter can remain effective for months or even years. Regular monitoring is vital to prevent complications such as infection or clotting.
Advanced Features
- Dual-Lumen Design
- Biocompatible Material
- Multiple Length Options
- Soft, Tapered Tip
- Long-Term Durability
- Complete Sterile Kit
- Multiple Length Options
Cardiovascular Excellence with PTFE Guide Wire Solutions
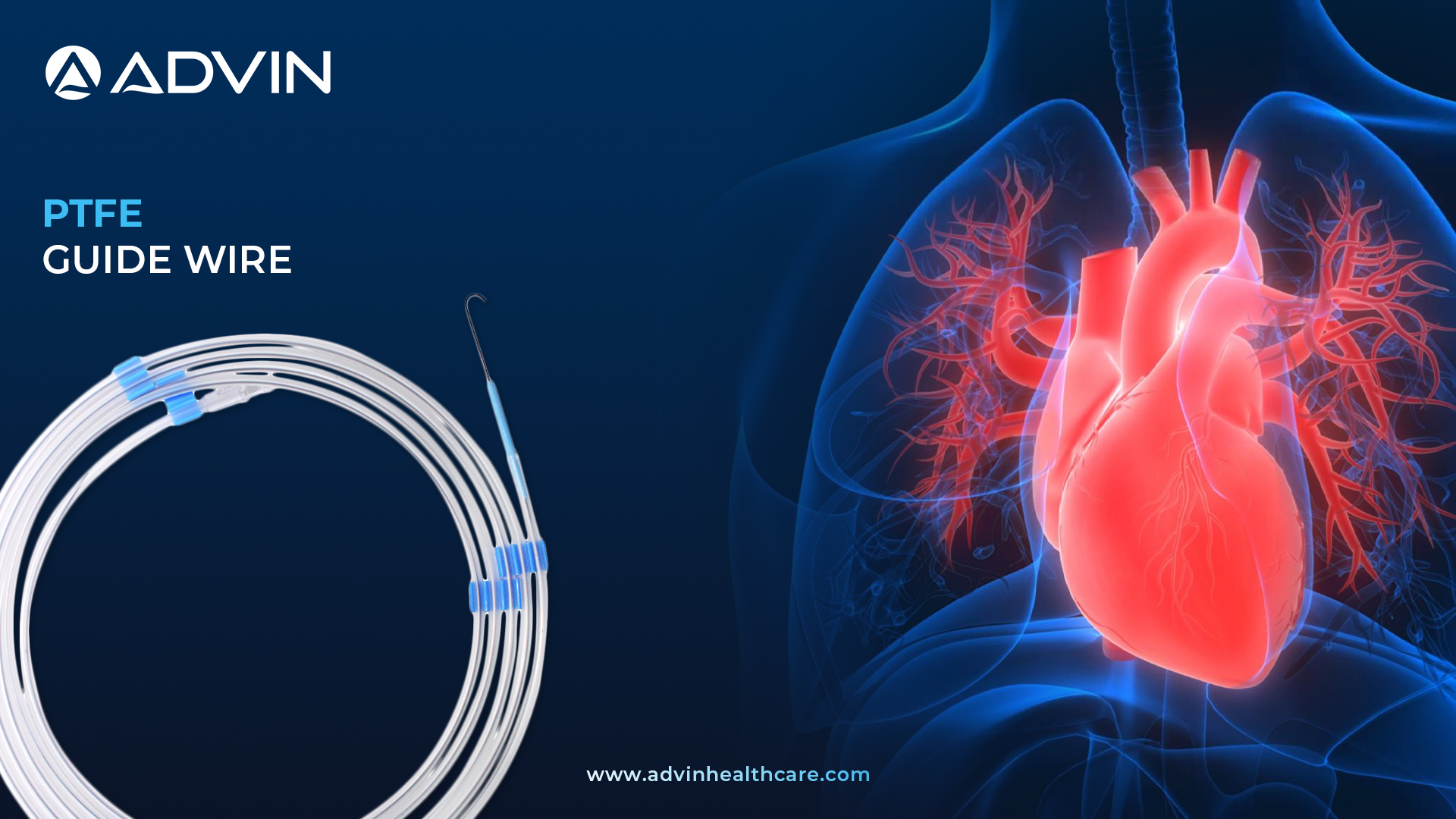
PTFE Guide Wire
PTFE Guide Wire is a medical device used in cardiology to help guide catheters or other instruments through blood vessels. It is coated with polytetrafluoroethylene (PTFE) to reduce friction and allow smooth navigation. Doctors commonly use this wire during procedures like angioplasty and catheterization.
Product History
PTFE Guide Wires were introduced to improve precision and reduce resistance during vascular procedures. Over the years, they became a standard tool in cardiology due to their excellent flexibility and smooth surface. The PTFE coating was developed to enhance glide and reduce trauma to blood vessels. Today, these wires are widely used in hospitals worldwide for various cardiovascular interventions.
PTFE Guide Wire – Low Friction, High Control in Every Procedure
Advin Health Care offers high-quality PTFE Guide Wires designed for efficient navigation through complex vascular pathways. These wires are coated with PTFE, which provides a smooth surface to reduce friction during insertion. The product is built for flexibility, strength, and easy handling, making it ideal for a wide range of cardiovascular procedures. Advin Health Care ensures strict quality control, resulting in safe and reliable performance during surgery. It is especially suitable for procedures requiring precise catheter placement in narrow or tortuous vessels.
Surgeries Related to Product
- Coronary angioplasty
- Cardiac catheterization
- Peripheral angioplasty
- Electrophysiology procedures
- Renal artery stenting
- Carotid artery stenting
- Structural heart interventions
- Endovascular aneurysm repair (EVAR)
- Venous interventions
- Balloon angioplasty
Instructions for Use
- Open the sterile package carefully.
- Select the appropriate guide wire size and type based on the procedure.
- Insert the PTFE Guide Wire slowly through the vascular access site.
- Advance the wire under fluoroscopic guidance to the desired location.
- Proceed with catheter or device insertion over the wire as needed.
Top 10 Countries of Surgeries
- United States
- China
- India
- Germany
- Japan
- Brazil
- United Kingdom
- France
- Italy
- South Korea
Advin PTFE Guide Wire Description
Advin Health Care is a prominent manufacturer of PTFE Guide Wires, offering precision-crafted devices that support safe and smooth vascular navigation in cardiology procedures.
Types of PTFE Guide Wires
Advin Health Care provides a wide range of PTFE Guide Wires to suit different clinical needs and surgical preferences.
1. Based on Coating Type
- PTFE Coated Guide Wire
- Hydrophilic Coated Guide Wire
- Hydrophobic Coated Guide Wire
- Non-Coated Guide Wire
2. Based on Tip Configuration
- Straight Tip Guide Wire
- Angled Tip Guide Wire
- J Tip Guide Wire
Craniotomy Drapes
Craniotomy Drapes
Craniotomy Drapes are sterile surgical drapes used during brain surgery to maintain a clean and infection-free area. These drapes are specially designed to cover the patient and expose only the surgical site on the head. They are used by neurosurgeons during craniotomy and other brain-related procedures.
Product History
Craniotomy Drapes have been developed to meet the specific needs of neurosurgical procedures. Over time, materials and designs have improved for better absorption and protection. The evolution of these drapes has focused on preventing surgical site infections and improving patient safety. Today, NIVDA Drapes produces high-quality Craniotomy Drapes trusted by hospitals worldwide.
Craniotomy Drapes – Advanced Barrier Protection for Complex Cranial Procedures
NIVDA Drapes Craniotomy Drapes are designed to provide a secure and sterile field during complex brain surgeries. They feature fluid-resistant materials that protect both patients and surgical staff. With reinforced fenestration and adhesive zones, the drapes help maintain positioning throughout long procedures. NIVDA Drapes ensure reliable performance under critical surgical conditions. These drapes also minimize the risk of contamination and reduce surgical preparation time
Surgeries Related to Product
Craniotomy Drapes are commonly used in neurosurgical procedures such as craniotomy, brain tumor removal, aneurysm repair, epilepsy surgery, and trauma-related brain surgeries.
Instructions for Use
- Open the sterile package in a clean, designated area.
- Identify the correct orientation and fenestration area for the surgical site.
- Drape over the patient, ensuring the head area is aligned properly.
- Use adhesive zones to secure the drape in place.
- Dispose of after single use, following hospital waste disposal protocols.
Top 10 Countries of Surgeries
- United States
- China
- India
- Germany
- Japan
- Brazil
- United Kingdom
- Russia
- France
- South Korea
NIVDA Product Description
NIVDA Drapes is a leading manufacturer of Craniotomy Drapes, offering safe, high-performance solutions for neurosurgical procedures in hospitals and operating rooms globally. Each drape is crafted using medical-grade, fluid-resistant materials that ensure superior protection and patient safety. NIVDA products are manufactured in ISO 13485-certified facilities and comply with CE standards, reflecting our commitment to international quality and regulatory excellence. Trusted by surgeons worldwide, NIVDA Craniotomy Drapes are known for their precision design, secure fit, and ability to maintain a sterile surgical environment. Continuous innovation, strict quality control, and adherence to global healthcare standards make NIVDA Drapes a preferred choice in advanced neurosurgical care.
OTIS Urethrotome - A Precision Instrument in Urethral Surgery
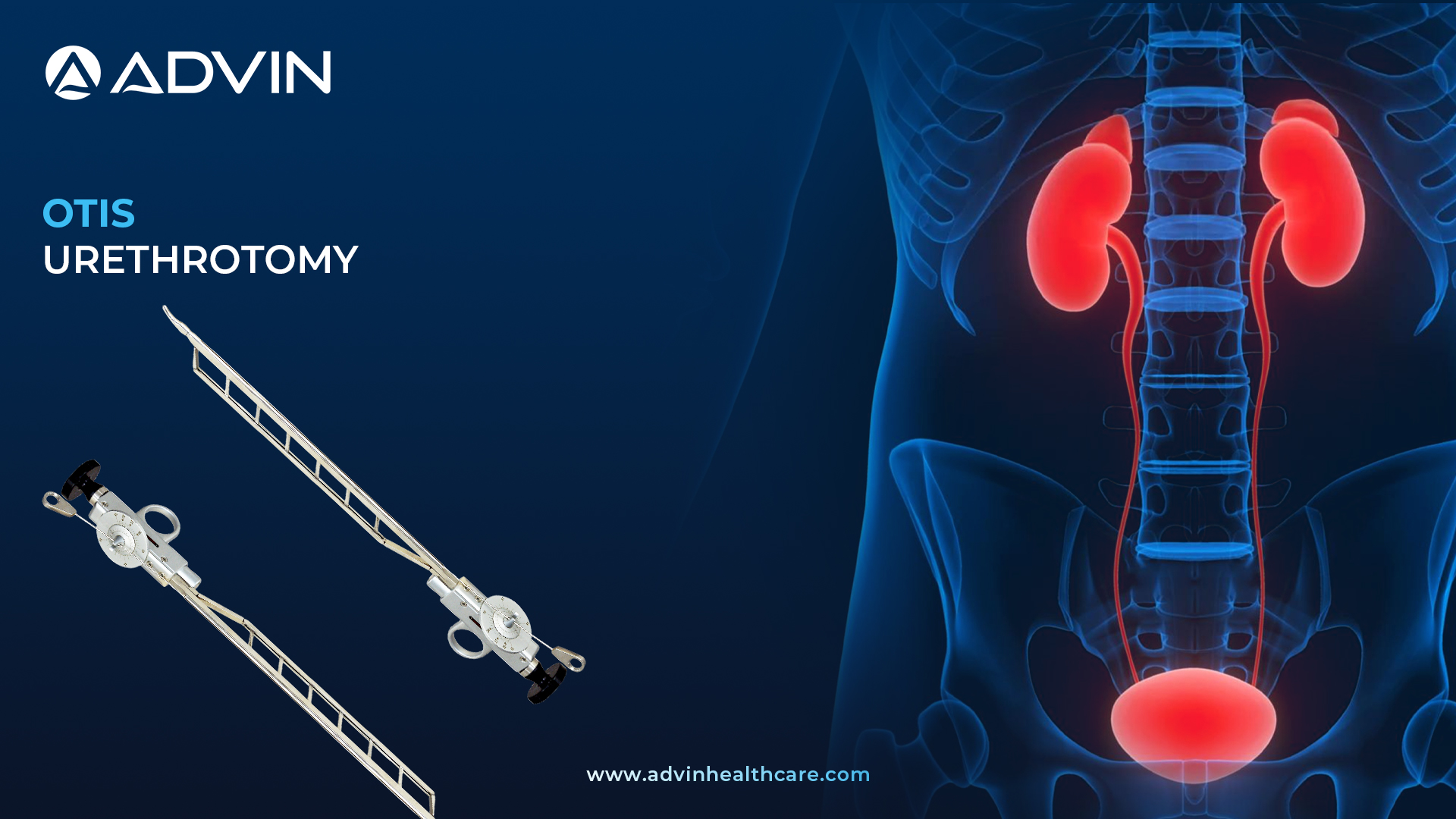
Introduction to the OTIS Urethrotome: A Precision Instrument in Urethral Surgery
The OTIS Urethrotome is a manual surgical instrument used to treat urethral strictures by cutting the narrowed section of the urethra. It provides controlled dilation and incision of the urethral passage. This instrument is widely used in urology for internal urethrotomy procedures.
Evolution of the OTIS Urethrotome: From Concept to Clinical Excellence
The OTIS Urethrotome was first developed in the 19th century by French surgeon Jean-François Otis, giving the device its name. It provided a safer alternative to blind dilation for urethral stricture treatment. Over time, the instrument has been refined to allow for more precise and adjustable incisions. It remains a reliable tool, especially in resource-limited settings or where visual urethrotomy is not possible.
What is the OTIS Urethrotome? A Concise Overview
The OTIS Urethrotome by Advin Health Care is a time-tested instrument used for internal urethral stricture treatment without the use of endoscopic visualization. Its adjustable blade allows surgeons to control the depth of incision while dilating the stricture. Advin Health Care ensures a smooth-operating design and ergonomic handle for better control during the procedure. This device is particularly helpful in treating anterior urethral strictures. It is easy to clean, reusable, and compatible with common urological instruments.
Surgical Applications of the OTIS Urethrotome: Enhancing Urethral Stricture Management
- Internal Urethrotomy (Non-Visual)
- Anterior Urethral Stricture Treatment
- Urethral Dilation and Cutting
- Pre-Endoscopic Stricture Management
How to Use the OTIS Urethrotome Effectively
Lubricate and prepare the OTIS Urethrotome before use. Gently insert the instrument into the urethra up to the location of the stricture. Adjust the cutting blade to the desired depth using the control mechanism. Slowly withdraw the instrument while performing the incision. Clean and sterilize the device after use.
Global Utilization: Top 10 Countries Advancing Urology with the OTIS Urethrotome
- India
- Egypt
- China
- Nigeria
- Brazil
- Pakistan
- Bangladesh
- Indonesia
- Kenya
- Philippines
Advin OTIS Urethrotome: Product Specifications and Features
Advin Health Care is a reliable manufacturer of OTIS Urethrotome, delivering robust, adjustable instruments for effective and economical urethral stricture management in diverse clinical settings.
An OTIS urethrotomy is the blind transurethral incision of the urethra by an Otis Urethrotome. After insertion of the Otis Urethrotome, the urethra is dilated to the desired width and a blade cuts the stretched urethra at the 12 o’clock position. OTIS Urethrotome is also called as Blind Internal Urethrotomy.
The Otis urethrotomy is reserved for the palliative treatment of long-segment strictures of the male urethra or to treat proximal narrowing of the female urethra. Otis urethrotomy is also performed before the use of old resectoscopes (> 26 CH) in a narrow urethra to prevent ischemic damage.
Advanced Features
- Designed to provide excellent utility
- Accurate dimensions
- Advanced configuration
- Anti-corrosive
- Smooth finish
- High performance
- Long lasting nature
Endoscopy Coagulation Forceps Explained - Features, Uses & Surgical Relevance
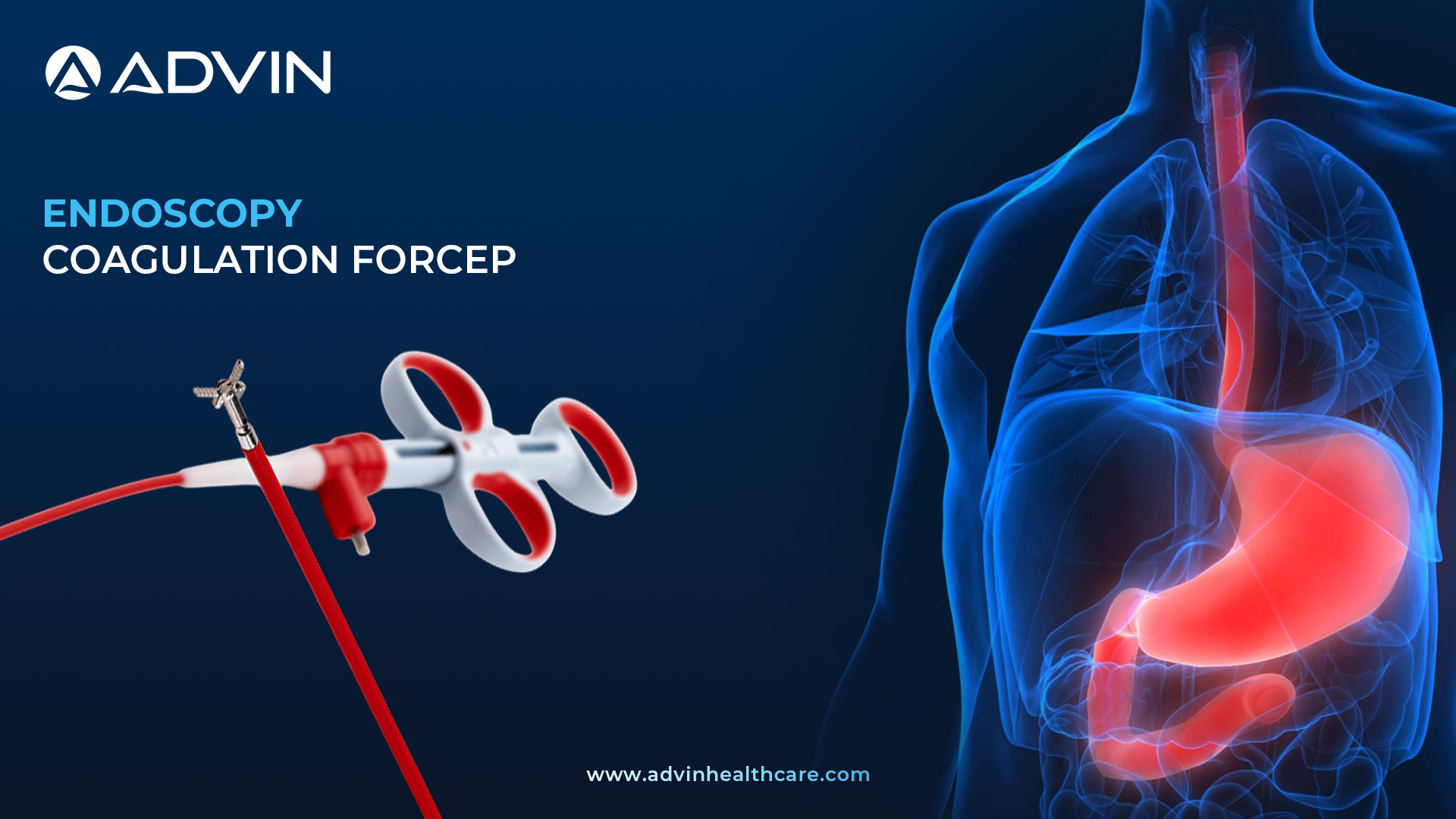
What is an Endoscopy Coagulation Forceps?
An Endoscopy Coagulation Forceps is a specialized medical instrument used during endoscopic procedures to stop bleeding inside the gastrointestinal (GI) tract. It works by applying a controlled electric current to cauterize tissue or blood vessels. This tool is commonly used in hospitals and endoscopy suites to manage bleeding and remove abnormal tissue.
The Evolution of Coagulation Forceps in Endoscopy
Coagulation forceps have been used in therapeutic endoscopy for several decades. Early designs were simple grasping tools, but modern versions are integrated with electrosurgical capabilities for better control. The development of high-frequency energy delivery systems improved their precision and safety. Over time, these forceps have become a standard tool in GI procedures worldwide.
Product Overview: Short Description
Advin Health Care offers high-quality Endoscopy Coagulation Forceps designed for precision and reliability in endoscopic interventions. These forceps allow physicians to safely cauterize bleeding sites and remove problematic tissue without causing unnecessary damage. The electrocoagulation tip provides targeted energy delivery for better outcomes. Advin Health Care ensures compatibility with most endoscopy systems, making the product highly versatile. This tool is single-use, sterile, and ergonomically designed for ease of handling during delicate procedures.
Endoscopy Coagulation Forceps are commonly used in:
- Gastrointestinal (GI) bleeding control
- Colonoscopic polypectomy
- Endoscopic Mucosal Resection (EMR)
- Treatment of angiodysplasia
- Esophageal variceal banding follow-up cauterization
How to Use Endoscopy Coagulation Forceps
- Connect the forcep to a compatible high-frequency generator if required.
- Insert the forcep through the working channel of the endoscope.
- Position the tip at the target bleeding site or lesion.
- Apply the electrical current as per clinical need to cauterize tissue.
- Withdraw and dispose of the device after a single use.
Clinical Benefits of Using Coagulation Forceps
Endoscopy Coagulation Forceps provide excellent hemostatic control during GI procedures. Their targeted energy application minimizes the risk of perforation and reduces procedure time. These forceps are essential in managing bleeding from ulcers, tumors, or post-polypectomy sites. Their compatibility with multiple endoscopic systems enhances workflow efficiency in surgical settings. The use of disposable, sterile instruments also reduces the risk of infection.
Advin Endoscopy Coagulation Forceps Description
Advin Health Care is a prominent manufacturer of Endoscopy Coagulation Forceps, offering dependable, sterile, and ergonomically designed instruments trusted by gastroenterologists and endoscopists globally. Each forcep is engineered to ensure precise electrocoagulation during complex GI procedures. With global distribution and a focus on safety, Advin sets the benchmark for medical device performance in therapeutic endoscopy.