EPO Injection – Boosting Red Blood Cell Production with Precision
EPO Injection
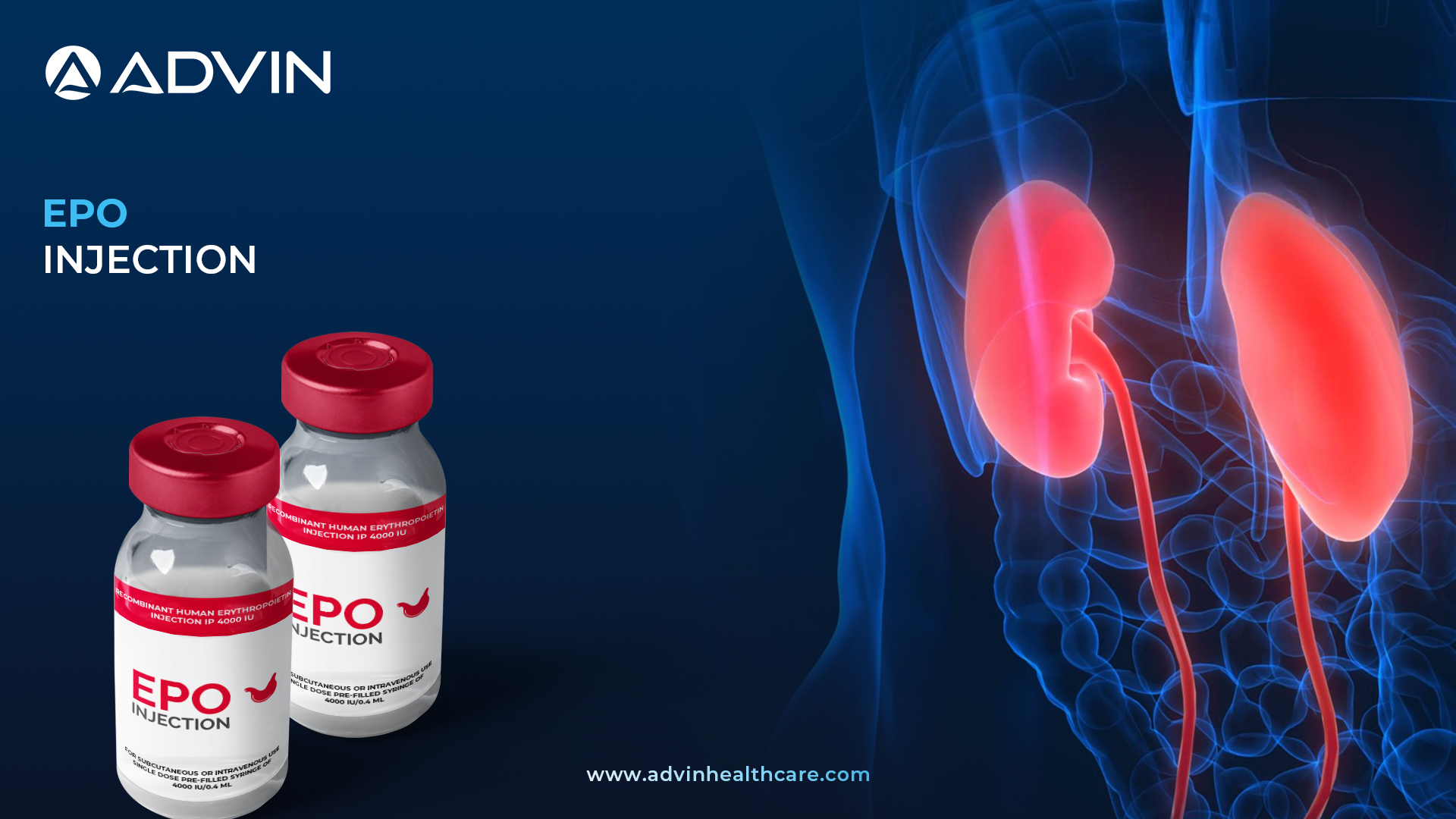
EPO Injection 4000IU is a medication used to treat anemia, especially in patients undergoing dialysis. It helps the body produce more red blood cells by stimulating the bone marrow. This injection is commonly used in hospitals and dialysis centers.
Product History
EPO, or erythropoietin, was first approved for medical use in the late 1980s. It was developed to help manage anemia in patients with chronic kidney disease. Over time, it became a standard treatment in dialysis to reduce the need for blood transfusions. The 4000IU dosage is one of the commonly used strengths in clinical settings.
Effective Support for Restoring Vital Hemoglobin Levels
EPO Injection 4000IU by Advin Health Care is used to treat anemia in patients with chronic kidney disease, especially those on dialysis. This injection helps increase red blood cell count, improving oxygen transport in the body. Advin Health Care ensures each dose is produced with high-quality standards and precise formulation. The 4000IU dosage is ideal for stable patients needing regular anemia management. It can reduce fatigue and improve quality of life in dialysis patients.
Surgeries Related to Product
- Hemodialysis procedures
- Peritoneal dialysis
- Kidney transplant surgeries
- Pre- and post-operative management in renal failure cases
- Anemia management during long-term dialysis treatment
Instructions for Use
- Shake the vial gently before use.
- Administer the injection under the skin (subcutaneous) or into a vein (intravenous), as advised by a doctor.
- Store the injection in a refrigerator between 2°C and 8°C.
- Do not freeze or shake vigorously.
- Always use under the supervision of a healthcare professional.
Top 10 Countries of Surgeries
- United States
- India
- China
- Germany
- Brazil
- Japan
- United Kingdom
- Russia
- Mexico
- Turkey
Advin Product Description
- Advin Health Care is a prominent manufacturer of EPO Injection 4000IU, offering reliable and high-quality solutions for anemia management in dialysis patients.
- EPO 4000IU INJECTION is a medicine that helps your bone marrow to produce more red blood cells. It is used to treat a type of anemia caused by kidney disease. It is also used to treat anemia caused by cancer chemotherapy and by taking medicines to treat HIV.
Mullin Introducer Sheath – Smooth, Reliable Access for Transseptal Procedures
Mullin Introducer Sheath
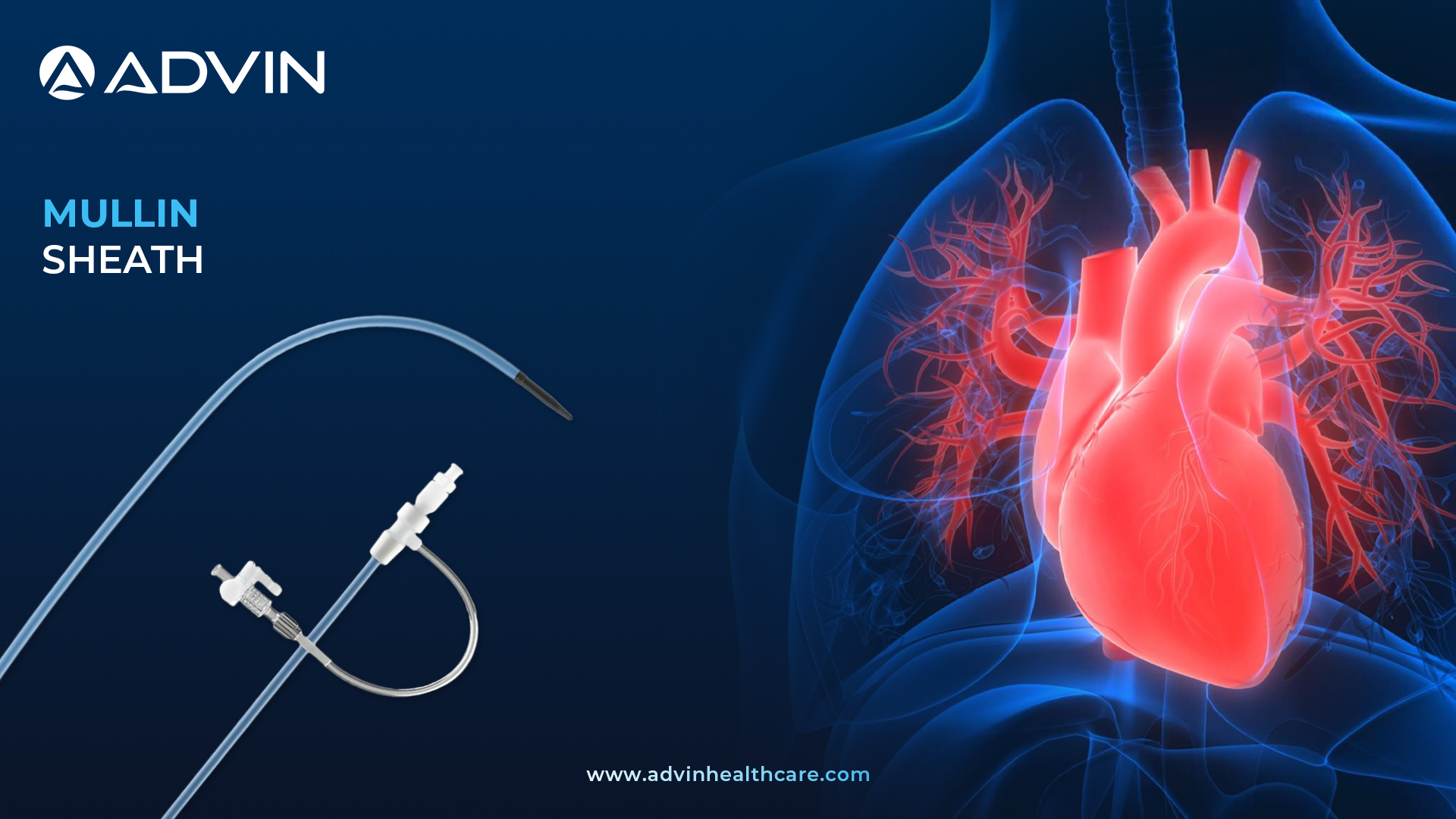
The Mullin Introducer Sheath is a medical device used in cardiology procedures to provide access to blood vessels. It helps guide catheters and other instruments into the heart during diagnostic and interventional procedures. The sheath is designed to minimize trauma and ensure safe insertion.
Product History
The Mullin Introducer Sheath was developed to support safer and more controlled access to the vascular system during heart procedures. It became widely used in cardiology after its effectiveness in minimizing vascular damage was proven. Over the years, improvements in material and design have enhanced its performance and patient safety. Today, it remains a key tool in cardiac catheterization labs around the world.
Engineered for Safety in Every Septal Puncture.
Advin Health Care provides high-quality Mullin Introducer Sheaths for advanced cardiology use. The sheath allows smooth insertion of catheters with reduced vessel trauma and enhanced stability. It is available in different sizes to suit various patient and procedural needs. Advin Health Care ensures the product is made from biocompatible materials with excellent flexibility and strength. This product is ideal for precise access in both routine and complex cardiac procedures.
Surgeries Related to Product
- Cardiac catheterization
- Electrophysiology studies
- Pacemaker and defibrillator lead placement
- Coronary angiography
- Radiofrequency ablation procedures
- Left heart access in structural heart interventions
Instructions for Use
- Select the appropriate size and type of Mullin Introducer Sheath.
- Prepare the insertion site using sterile technique.
- Insert the sheath into the vessel using a guidewire.
- Secure the sheath in place and confirm placement.
- Introduce diagnostic or therapeutic instruments through the sheath as needed.
Top 10 Countries of Surgeries
- United States
- India
- China
- Germany
- Brazil
- United Kingdom
- Japan
- Russia
- France
- South Korea
Advin Product Description
Advin Health Care is a leading manufacturer of Mullin Introducer Sheath, offering a safe and efficient solution for vascular access in cardiology procedures with international quality standards.
Enhanced Features of Mullin Introducer Sheath
- Radiopaque Sheath Design
- High Kink Resistance
- Low-Resistance Smooth Taper
- Precision-Enhanced Tip
- Sterilized with Ethylene Oxide (ETO)
- Individually Packed, Single-Use Only
- 67 cm Dilator Length
- Guide Wire Compatibility: 0.032″
Stone Crushing Forceps - Highly Prefers For Bladder Stone Removal
Introduction to Stone Crushing Forceps: Precision Tools for Endoscopic Stone Management:
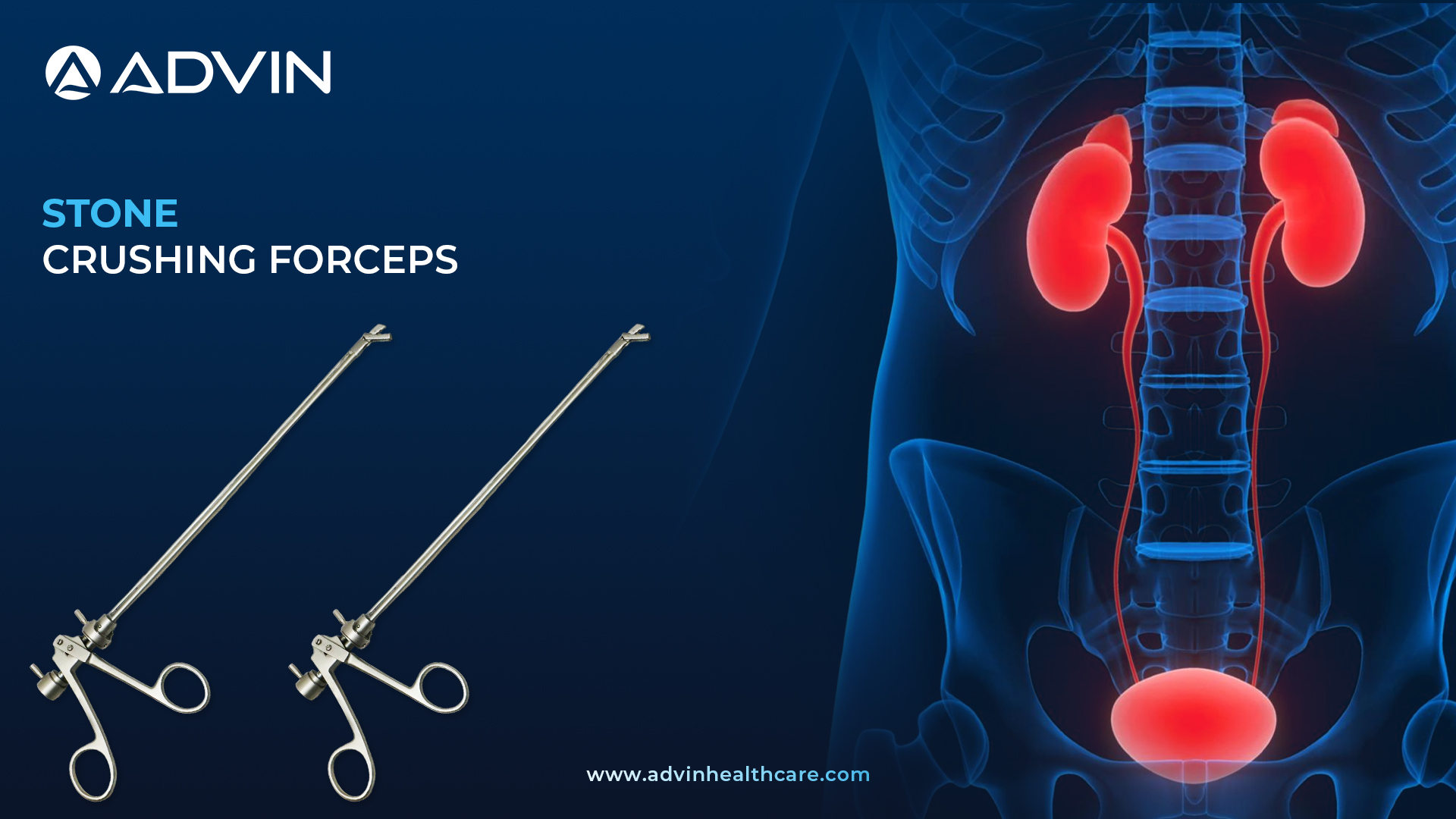
Stone Crushing Forceps are specialized instruments used to break and remove bladder stones during cystoscopy procedures. They are inserted through a Cystoscope and operated to crush stones mechanically. This device allows urologists to manage bladder stones without open surgery.
Evolution of Stone Crushing Forceps: Advancing Urological Stone Fragmentation:
Bladder stones have been treated for centuries, initially with open surgical techniques. The invention of stone crushing forceps allowed for endoscopic access to the bladder, reducing complications and recovery time. Over time, the instrument evolved to become more ergonomic and efficient, with improved designs for crushing power and precision. Today, it remains a vital tool in minimally invasive urology procedures.
What Are Stone Crushing Forceps? A Brief Overview for Clinical Use:
The Stone Crushing Forceps by Advin Health Care are designed for the safe and effective fragmentation of bladder stones under direct endoscopic vision. These forceps are compatible with standard cystoscopes and offer strong grip and crushing capability. Advin Health Care uses high-quality, medical-grade stainless steel to ensure reliability and durability during repeated use. The ergonomic handle design provides excellent control and user comfort. It is an essential tool for urologists performing stone management without energy sources.
Surgical Applications: Effective Use of Stone Crushing Forceps in Lithotripsy Procedures:
- Bladder Stone Removal
- Cystolitholapaxy
- Endoscopic Bladder Stone Fragmentation
- Transurethral Stone Extraction Procedures
How to Use Stone Crushing Forceps:
Assemble the forceps with the Cystoscope and insert into the bladder through the urethra. Locate the stone under direct vision. Open the forceps jaws, position around the stone, and gently close to crush it. Repeat as needed until fragments are small enough to remove. Clean and sterilize thoroughly after each use.
Global Trends: Top 10 Countries Utilizing Stone Crushing Forceps in Endourology:
- India
- Egypt
- China
- Pakistan
- Nigeria
- Indonesia
- Bangladesh
- Brazil
- United States
- Turkey
Advin Stone Crushing Forceps: Product Features, Build Quality & Technical Specifications:
- Advin Health Care is a prominent manufacturer of Stone Crushing Forceps, delivering precision instruments for effective and minimally invasive bladder stone removal during cystoscopy procedures.
- Stone Crushing Forceps is use to remove bladder stone. It is compatible with 23.5 Fr or 25 Fr Cystoscopy Sheath. 4mm 30 Degree telescope will be attached with Stone Crushing Forceps.
- Stone Crushing Forceps also called as Gall bladder Stone Crushing Equipment, Urology Stone Crushing Forceps.
Advanced Features:
- 1 Provide excellent utility
- Advanced configuration
- Autoclavable
- Highly Compatible
- Compatible with Karl STORZ and Richard Wolf
Laparotomy Drapes
Laparotomy Drapes
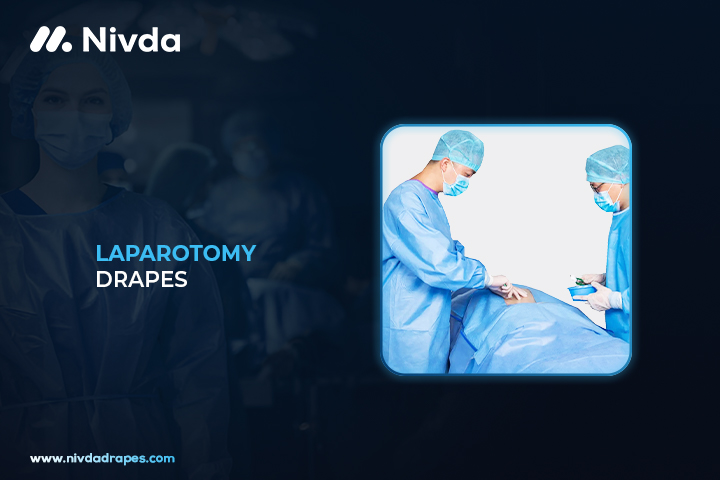
Laparotomy Drapes are specialized sterile covers used during abdominal surgeries to maintain a clean surgical field. They are made from high-quality, fluid-resistant material that protects both the patient and surgical staff. These drapes are part of the NIVDA Drapes product range used widely in hospitals and surgical centers.
Product History
Laparotomy Drapes have been in use for many decades and have evolved with improvements in surgical techniques and materials. Initially, simple cloths were used, but modern drapes are now made from advanced, non-woven, disposable materials. NIVDA Drapes introduced its laparotomy drapes to provide enhanced infection control and ease of use. Over time, they became one of the preferred choices for abdominal procedures in many countries.
Laparotomy Drapes – Designed for Maximum Protection and Fluid Control
NIVDA Drapes Laparotomy Drapes are designed for abdominal surgeries, providing maximum coverage and fluid control. These drapes feature adhesive strips and fluid collection pouches to reduce contamination risks. NIVDA Drapes are manufactured using breathable, tear-resistant fabric to ensure patient comfort and surgical efficiency. They are easy to position and stay securely in place during the procedure. The product helps reduce surgical site infections by maintaining a sterile environment around the incision area.
Common Surgeries Where Laparotomy Drapes Are Used
Exploratory laparotomy
Abdominal tumor removal
Intestinal surgeries (e.g., resection, obstruction relief)
Appendectomy (open)
Hysterectomy (abdominal)
Liver and spleen surgeries
Emergency trauma laparotomies
Instructions for Use
- Ensure the patient is properly positioned on the operating table.
- Open the sterile laparotomy drape pack using aseptic technique.
- Drape the patient starting from the incision site outward.
- Secure adhesive edges to keep the drape in place.
- Adjust fluid collection pouch if available to manage fluids during surgery.
Customization Options for Different Surgical Needs
NIVDA Laparotomy Drapes are available in multiple configurations to suit various abdominal procedures, from standard exploratory surgeries to complex oncologic or trauma cases. Hospitals can choose from options with or without fluid pouches, varying fenestration sizes, and added features like tubing holders or arm board covers—ensuring each surgical team gets exactly what they need for optimal performance.
Enhanced Patient Safety through Advanced Barrier Protection
These drapes are engineered with a high-performance barrier layer that blocks microbial and fluid transmission, reducing the risk of surgical site infections (SSIs). The fabric maintains a balance of strength and breathability, which not only protects the patient but also helps maintain a comfortable environment in the sterile field for surgeons and staff.
NIVDA Laparotomy Drapes Description
NIVDA Drapes is a leading manufacturer of Laparotomy Drapes, offering high-quality, sterile surgical solutions that enhance safety and infection control during abdominal procedures. Each drape is made from reinforced, non-woven SMS (Spunbond-Meltblown-Spunbond) fabric that is fluid-resistant and breathable. The standard dimensions range from 200 cm x 300 cm to 250 cm x 340 cm, with a central fenestration to allow precise access to the surgical site. NIVDA Laparotomy Drapes include integrated fluid collection pouches, adhesive edges, and instrument holders for added convenience during surgery. These drapes are EO-sterilized, individually packed, and meet ISO 13485 and CE certification standards to ensure the highest level of quality and compliance.
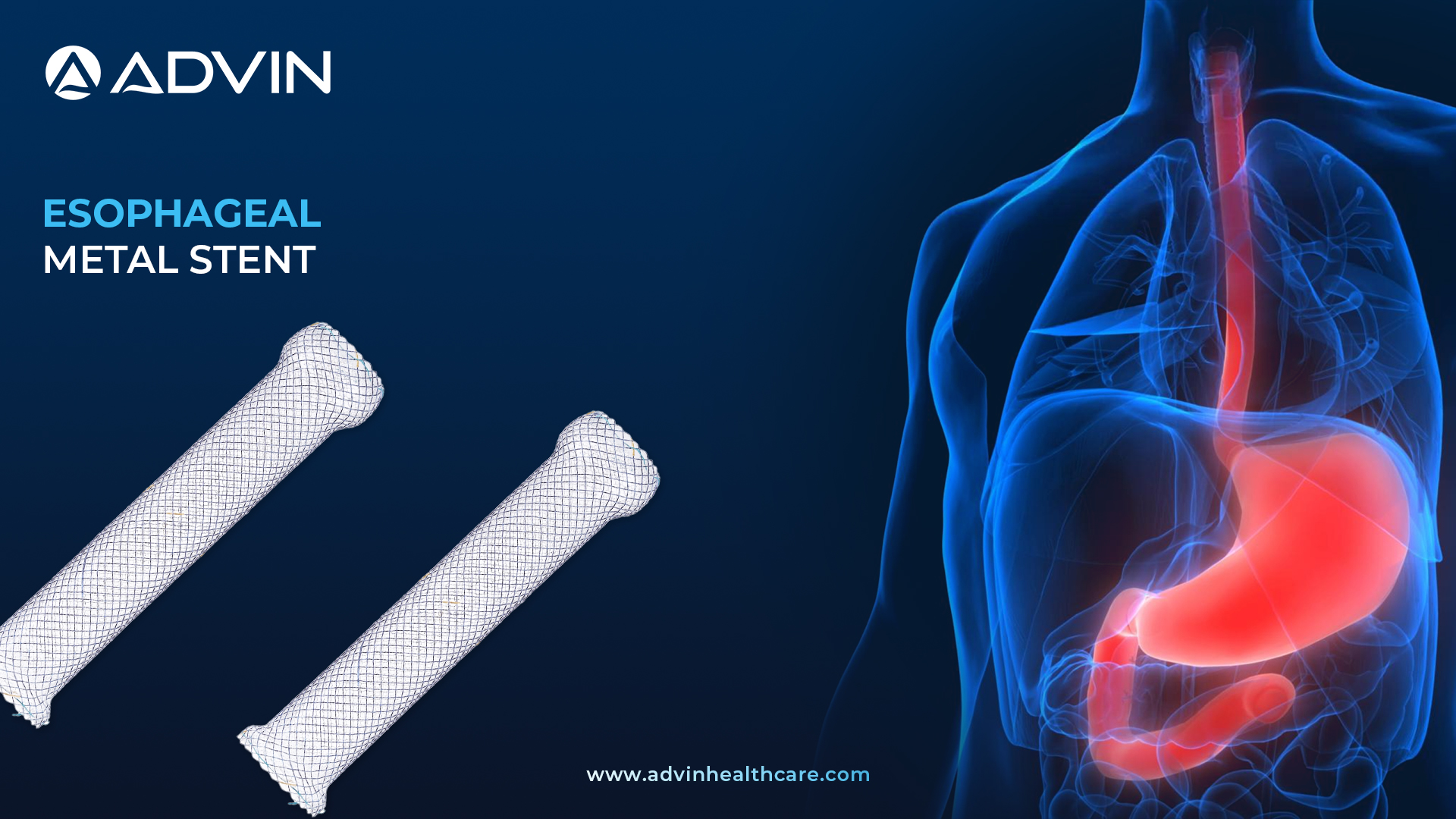
Esophageal Metal Stents - Restoring Swallowing and Enhancing Patient Care
Esophageal Metal Stents
The Esophageal Metal Stent is a specialized medical device used to keep the esophagus open in patients with strictures or blockages. It is designed with a strong, flexible metal structure that adapts to the esophageal shape. The stent is available in fully, partially, and uncovered types for various clinical needs.
Product Evolution and Development
The idea of using stents in the esophagus has been around for many years, with initial models being fairly basic. Major progress was made in the late 1900s with the introduction of self-expanding metal stents (SEMS), which improved both flexibility and placement techniques. Since then, newer designs with covering options and better materials have been developed to improve results and minimize risks. These continuous improvements aim to make treatment more effective and comfortable for patients.
Why Choose Advin Health Care’s Esophageal Metal Stents?
Advin Health Care offers Esophageal Metal Stents that combine clinical reliability with patient-centered design. These stents are fully coated with silicone to help prevent tumor ingrowth and reduce food blockage, making them suitable for long-term use. The structure supports repositioning during placement, giving clinicians more control and accuracy. With flared ends and optimal radial force, the stent remains secure while effectively expanding the esophageal passage. Advin Health Care ensures each stent features a hook-and-cross design that allows gradual expansion for a smooth and comfortable fit.
Common Surgical Applications
The Esophageal Metal Stent is commonly used in:
- Esophageal cancer surgery
- Palliative care for malignant strictures
- Management of benign esophageal strictures
- Treatment of esophageal fistulas
- Post-surgical esophageal narrowing
Basic Instructions for Use
- Choose the appropriate stent size based on patient anatomy.
- Insert the stent over a guidewire using endoscopic guidance.
- Gradually deploy the stent while monitoring placement.
- Use the retrieval lasso for repositioning if required.
- Confirm final positioning with fluoroscopic or endoscopic imaging.
Choosing the Right Esophageal Stent: Fully vs. Partially Covered
The choice between fully and partially covered esophageal stents depends on the patient’s condition and treatment goal. Fully covered stents help prevent tumor ingrowth and are easier to reposition or remove. Partially covered stents offer stronger anchorage and reduce the risk of migration. The final selection is made by the healthcare provider based on clinical needs.
Advin Esophageal Metal Stents Description
Advin Health Care is a prominent manufacturer of Esophageal Metal Stents, offering solutions that meet both clinical precision and patient comfort. These stents are available in a wide range of lengths and designs to suit diverse anatomical and therapeutic requirements. Built with superior materials and engineering, Advin’s stents are trusted by healthcare providers globally.