Stone Crushing Forceps - Highly Prefers For Bladder Stone Removal
Introduction to Stone Crushing Forceps: Precision Tools for Endoscopic Stone Management:
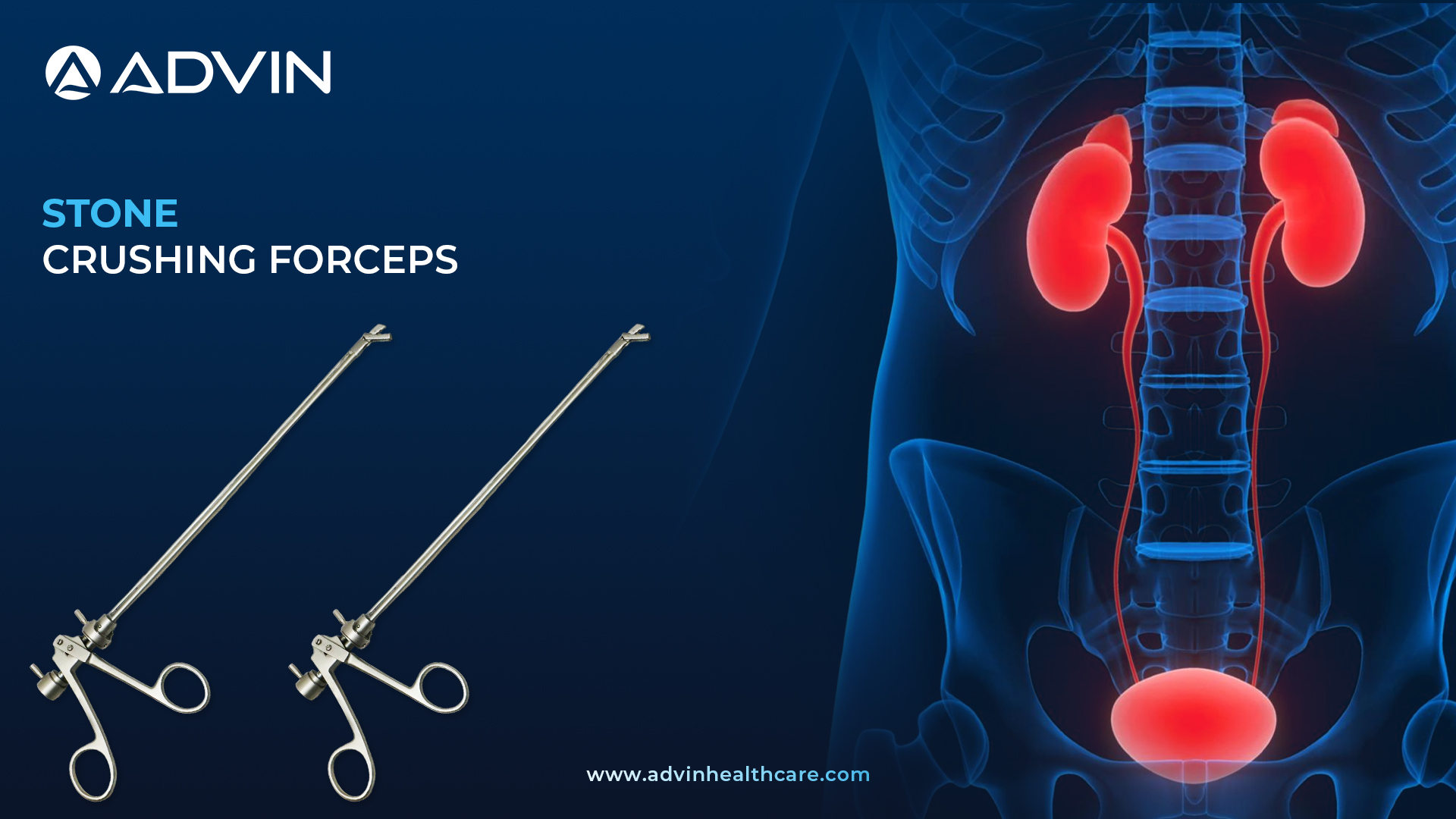
Stone Crushing Forceps are specialized instruments used to break and remove bladder stones during cystoscopy procedures. They are inserted through a Cystoscope and operated to crush stones mechanically. This device allows urologists to manage bladder stones without open surgery.
Evolution of Stone Crushing Forceps: Advancing Urological Stone Fragmentation:
Bladder stones have been treated for centuries, initially with open surgical techniques. The invention of stone crushing forceps allowed for endoscopic access to the bladder, reducing complications and recovery time. Over time, the instrument evolved to become more ergonomic and efficient, with improved designs for crushing power and precision. Today, it remains a vital tool in minimally invasive urology procedures.
What Are Stone Crushing Forceps? A Brief Overview for Clinical Use:
The Stone Crushing Forceps by Advin Health Care are designed for the safe and effective fragmentation of bladder stones under direct endoscopic vision. These forceps are compatible with standard cystoscopes and offer strong grip and crushing capability. Advin Health Care uses high-quality, medical-grade stainless steel to ensure reliability and durability during repeated use. The ergonomic handle design provides excellent control and user comfort. It is an essential tool for urologists performing stone management without energy sources.
Surgical Applications: Effective Use of Stone Crushing Forceps in Lithotripsy Procedures:
- Bladder Stone Removal
- Cystolitholapaxy
- Endoscopic Bladder Stone Fragmentation
- Transurethral Stone Extraction Procedures
How to Use Stone Crushing Forceps:
Assemble the forceps with the Cystoscope and insert into the bladder through the urethra. Locate the stone under direct vision. Open the forceps jaws, position around the stone, and gently close to crush it. Repeat as needed until fragments are small enough to remove. Clean and sterilize thoroughly after each use.
Global Trends: Top 10 Countries Utilizing Stone Crushing Forceps in Endourology:
- India
- Egypt
- China
- Pakistan
- Nigeria
- Indonesia
- Bangladesh
- Brazil
- United States
- Turkey
Advin Stone Crushing Forceps: Product Features, Build Quality & Technical Specifications:
- Advin Health Care is a prominent manufacturer of Stone Crushing Forceps, delivering precision instruments for effective and minimally invasive bladder stone removal during cystoscopy procedures.
- Stone Crushing Forceps is use to remove bladder stone. It is compatible with 23.5 Fr or 25 Fr Cystoscopy Sheath. 4mm 30 Degree telescope will be attached with Stone Crushing Forceps.
- Stone Crushing Forceps also called as Gall bladder Stone Crushing Equipment, Urology Stone Crushing Forceps.
Advanced Features:
- 1 Provide excellent utility
- Advanced configuration
- Autoclavable
- Highly Compatible
- Compatible with Karl STORZ and Richard Wolf
Laparotomy Drapes
Laparotomy Drapes
Laparotomy Drapes are specialized sterile covers used during abdominal surgeries to maintain a clean surgical field. They are made from high-quality, fluid-resistant material that protects both the patient and surgical staff. These drapes are part of the NIVDA Drapes product range used widely in hospitals and surgical centers.
Product History
Laparotomy Drapes have been in use for many decades and have evolved with improvements in surgical techniques and materials. Initially, simple cloths were used, but modern drapes are now made from advanced, non-woven, disposable materials. NIVDA Drapes introduced its laparotomy drapes to provide enhanced infection control and ease of use. Over time, they became one of the preferred choices for abdominal procedures in many countries.
Laparotomy Drapes – Designed for Maximum Protection and Fluid Control
NIVDA Drapes Laparotomy Drapes are designed for abdominal surgeries, providing maximum coverage and fluid control. These drapes feature adhesive strips and fluid collection pouches to reduce contamination risks. NIVDA Drapes are manufactured using breathable, tear-resistant fabric to ensure patient comfort and surgical efficiency. They are easy to position and stay securely in place during the procedure. The product helps reduce surgical site infections by maintaining a sterile environment around the incision area.
Common Surgeries Where Laparotomy Drapes Are Used
Exploratory laparotomy
Abdominal tumor removal
Intestinal surgeries (e.g., resection, obstruction relief)
Appendectomy (open)
Hysterectomy (abdominal)
Liver and spleen surgeries
Emergency trauma laparotomies
Instructions for Use
- Ensure the patient is properly positioned on the operating table.
- Open the sterile laparotomy drape pack using aseptic technique.
- Drape the patient starting from the incision site outward.
- Secure adhesive edges to keep the drape in place.
- Adjust fluid collection pouch if available to manage fluids during surgery.
Customization Options for Different Surgical Needs
NIVDA Laparotomy Drapes are available in multiple configurations to suit various abdominal procedures, from standard exploratory surgeries to complex oncologic or trauma cases. Hospitals can choose from options with or without fluid pouches, varying fenestration sizes, and added features like tubing holders or arm board covers—ensuring each surgical team gets exactly what they need for optimal performance.
Enhanced Patient Safety through Advanced Barrier Protection
These drapes are engineered with a high-performance barrier layer that blocks microbial and fluid transmission, reducing the risk of surgical site infections (SSIs). The fabric maintains a balance of strength and breathability, which not only protects the patient but also helps maintain a comfortable environment in the sterile field for surgeons and staff.
NIVDA Laparotomy Drapes Description
NIVDA Drapes is a leading manufacturer of Laparotomy Drapes, offering high-quality, sterile surgical solutions that enhance safety and infection control during abdominal procedures. Each drape is made from reinforced, non-woven SMS (Spunbond-Meltblown-Spunbond) fabric that is fluid-resistant and breathable. The standard dimensions range from 200 cm x 300 cm to 250 cm x 340 cm, with a central fenestration to allow precise access to the surgical site. NIVDA Laparotomy Drapes include integrated fluid collection pouches, adhesive edges, and instrument holders for added convenience during surgery. These drapes are EO-sterilized, individually packed, and meet ISO 13485 and CE certification standards to ensure the highest level of quality and compliance.
Esophageal Metal Stents - Restoring Swallowing and Enhancing Patient Care
Esophageal Metal Stents
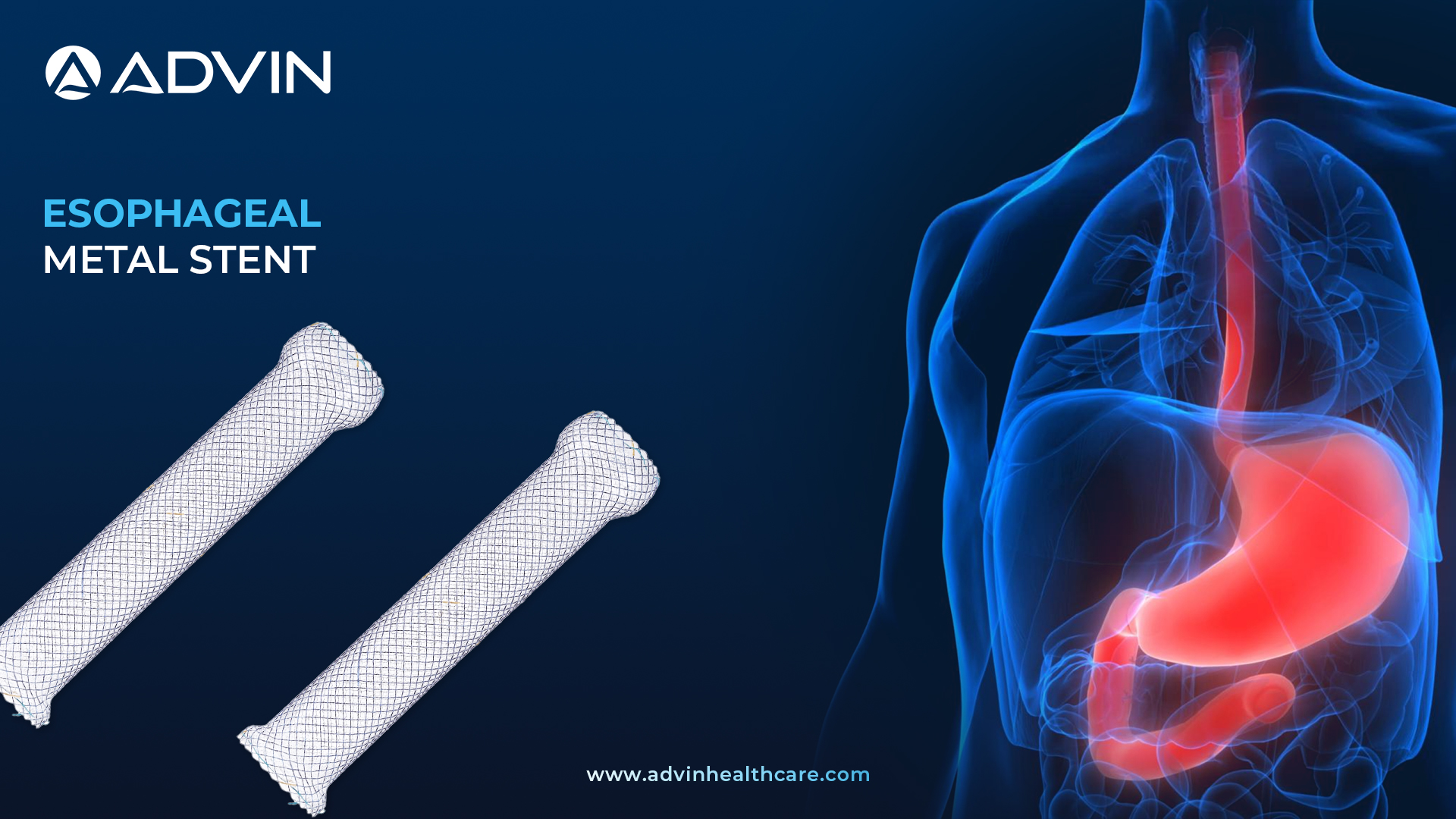
The Esophageal Metal Stent is a specialized medical device used to keep the esophagus open in patients with strictures or blockages. It is designed with a strong, flexible metal structure that adapts to the esophageal shape. The stent is available in fully, partially, and uncovered types for various clinical needs.
Product Evolution and Development
The idea of using stents in the esophagus has been around for many years, with initial models being fairly basic. Major progress was made in the late 1900s with the introduction of self-expanding metal stents (SEMS), which improved both flexibility and placement techniques. Since then, newer designs with covering options and better materials have been developed to improve results and minimize risks. These continuous improvements aim to make treatment more effective and comfortable for patients.
Why Choose Advin Health Care’s Esophageal Metal Stents?
Advin Health Care offers Esophageal Metal Stents that combine clinical reliability with patient-centered design. These stents are fully coated with silicone to help prevent tumor ingrowth and reduce food blockage, making them suitable for long-term use. The structure supports repositioning during placement, giving clinicians more control and accuracy. With flared ends and optimal radial force, the stent remains secure while effectively expanding the esophageal passage. Advin Health Care ensures each stent features a hook-and-cross design that allows gradual expansion for a smooth and comfortable fit.
Common Surgical Applications
The Esophageal Metal Stent is commonly used in:
- Esophageal cancer surgery
- Palliative care for malignant strictures
- Management of benign esophageal strictures
- Treatment of esophageal fistulas
- Post-surgical esophageal narrowing
Basic Instructions for Use
- Choose the appropriate stent size based on patient anatomy.
- Insert the stent over a guidewire using endoscopic guidance.
- Gradually deploy the stent while monitoring placement.
- Use the retrieval lasso for repositioning if required.
- Confirm final positioning with fluoroscopic or endoscopic imaging.
Choosing the Right Esophageal Stent: Fully vs. Partially Covered
The choice between fully and partially covered esophageal stents depends on the patient’s condition and treatment goal. Fully covered stents help prevent tumor ingrowth and are easier to reposition or remove. Partially covered stents offer stronger anchorage and reduce the risk of migration. The final selection is made by the healthcare provider based on clinical needs.
Advin Esophageal Metal Stents Description
Advin Health Care is a prominent manufacturer of Esophageal Metal Stents, offering solutions that meet both clinical precision and patient comfort. These stents are available in a wide range of lengths and designs to suit diverse anatomical and therapeutic requirements. Built with superior materials and engineering, Advin’s stents are trusted by healthcare providers globally.
Shoulder U Drapes
Shoulder U Drapes
Shoulder U Drapes are sterile surgical drapes used to cover and protect the patient’s body during shoulder surgeries. These drapes are shaped in a “U” form to allow better access to the shoulder area. They help maintain a sterile field and reduce the risk of infection during procedures.
Product History
Shoulder U Drapes were developed to meet the growing need for focused and reliable coverage in orthopaedic surgeries, especially shoulder operations. Over time, these drapes were improved with advanced barrier materials and better adhesive designs. Hospitals and surgical centers began adopting them for their practical shape and effective patient coverage. Today, they are used in many parts of the world as a trusted solution in sterile surgical practices.
Shoulder U Drapes – Designed for Comfort, Built for Performance
NIVDA Drapes offers Shoulder U Drapes designed specifically for shoulder procedures where clear access and full coverage are essential. These drapes have a U-shaped opening that helps isolate the surgical site while protecting surrounding areas. NIVDA Drapes are made with high-quality, fluid-resistant materials that support infection control. The drape includes adhesive borders that secure it firmly to the skin without slipping during surgery. Its smart design improves surgical efficiency and patient safety.
Surgeries Related to Product
Shoulder U Drapes are commonly used in surgeries such as shoulder arthroscopy, rotator cuff repair, shoulder replacement, and clavicle surgeries.
Instructions for Use
- Open the sterile drape packaging carefully.
- Position the drape over the patient’s shoulder area, aligning the U-shaped opening with the surgical site.
- Gently press the adhesive edges to secure the drape in place.
- Ensure the rest of the drape covers the surrounding areas to maintain sterility.
- Dispose of the drape after single use following standard hospital waste procedures.
Top 10 Countries of Surgeries
- United States
- Germany
- India
- China
- United Kingdom
- Brazil
- Japan
- Canada
- France
- Australia
NIVDA Product Description
NIVDA Drapes is a leading manufacturer of Shoulder U Drapes, providing high-quality sterile draping solutions for shoulder surgeries with optimal design, safety, and performance. These drapes are made from SMS (Spunbond-Meltblown-Spunbond) nonwoven fabric, known for its excellent fluid resistance and breathability. Each Shoulder U Drape features a U-shaped fenestration, integrated fluid collection pouch, and strong adhesive edges for secure placement. All NIVDA Drapes are EO sterilized, latex-free, and packed in easy-to-open sterile pouches for single use. Designed for efficiency and infection control, NIVDA Shoulder U Drapes meet international surgical draping standards and are trusted by hospitals worldwide.
Iron Sucrose Injection – Safe and Effective Iron Therapy for Better Patient Outcomes
Iron Sucrose Injection
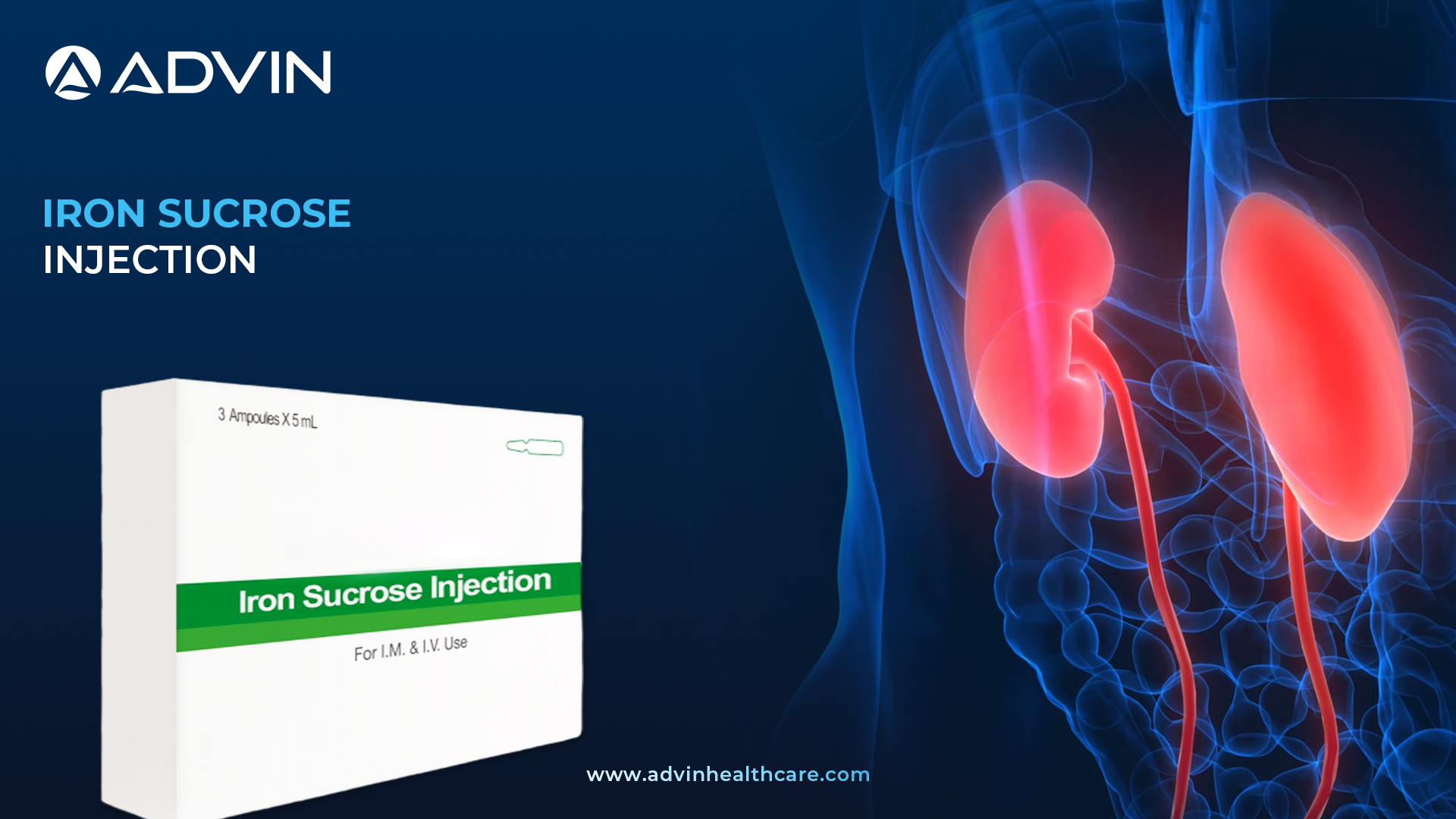
Iron Sucrose Injection is a sterile solution used to treat iron deficiency in patients undergoing dialysis. It helps increase iron levels in the blood, which is essential for making healthy red blood cells. This product is commonly used in hospitals to support patients with chronic kidney disease.
Product History
Iron Sucrose Injection was introduced as an effective treatment to manage iron deficiency anemia in patients with kidney problems. It offered an alternative to oral iron supplements, which are less effective in dialysis patients. Over time, it became widely accepted due to its safety and fast action. Today, it is a standard treatment in many dialysis centers worldwide.
Rapid Iron Replenishment for Better Patient Outcomes
Advin Health Care provides Iron Sucrose Injection designed to improve iron levels safely in dialysis patients. This product by Advin Health Care helps in managing anemia effectively by replenishing iron stores directly in the bloodstream. Iron Sucrose Injection reduces the need for blood transfusions, lowering associated risks. It works quickly to improve energy and overall well-being in patients with iron deficiency. The injection is simple to administer and well tolerated in hospital settings.
Surgeries Related to Product
Iron Sucrose Injection is commonly used in surgeries related to chronic kidney disease treatment, such as hemodialysis and peritoneal dialysis procedures. It is also used during kidney transplant preparations and other vascular access surgeries related to dialysis.
Instructions for Use
Iron Sucrose Injection should be administered intravenously by a healthcare professional. The dosage depends on the patient’s iron levels and overall health condition. It is typically given in small doses over a few minutes during or after dialysis sessions. Patients should be monitored for any allergic reactions during the injection. Follow the healthcare provider’s advice on the frequency and amount of injection.
Top 10 Countries of Surgeries
- United States
- Germany
- Japan
- Brazil
- India
- Canada
- United Kingdom
- France
- Australia
- South Korea
Advin Product Description
- Advin Health Care is a leading manufacturer of Iron Sucrose Injection, providing high-quality products that support patients with iron deficiency during dialysis. Their Iron Sucrose Injection is trusted in many hospitals for its safety, effectiveness, and ease of use in managing anemia associated with kidney disease.
- Iron sucrose injection is an iron replacement product that is used to treat iron deficiency anemia (not enough iron in the blood) in patients with chronic kidney disease (CKD). Iron is a mineral that the body needs to produce red blood cells.
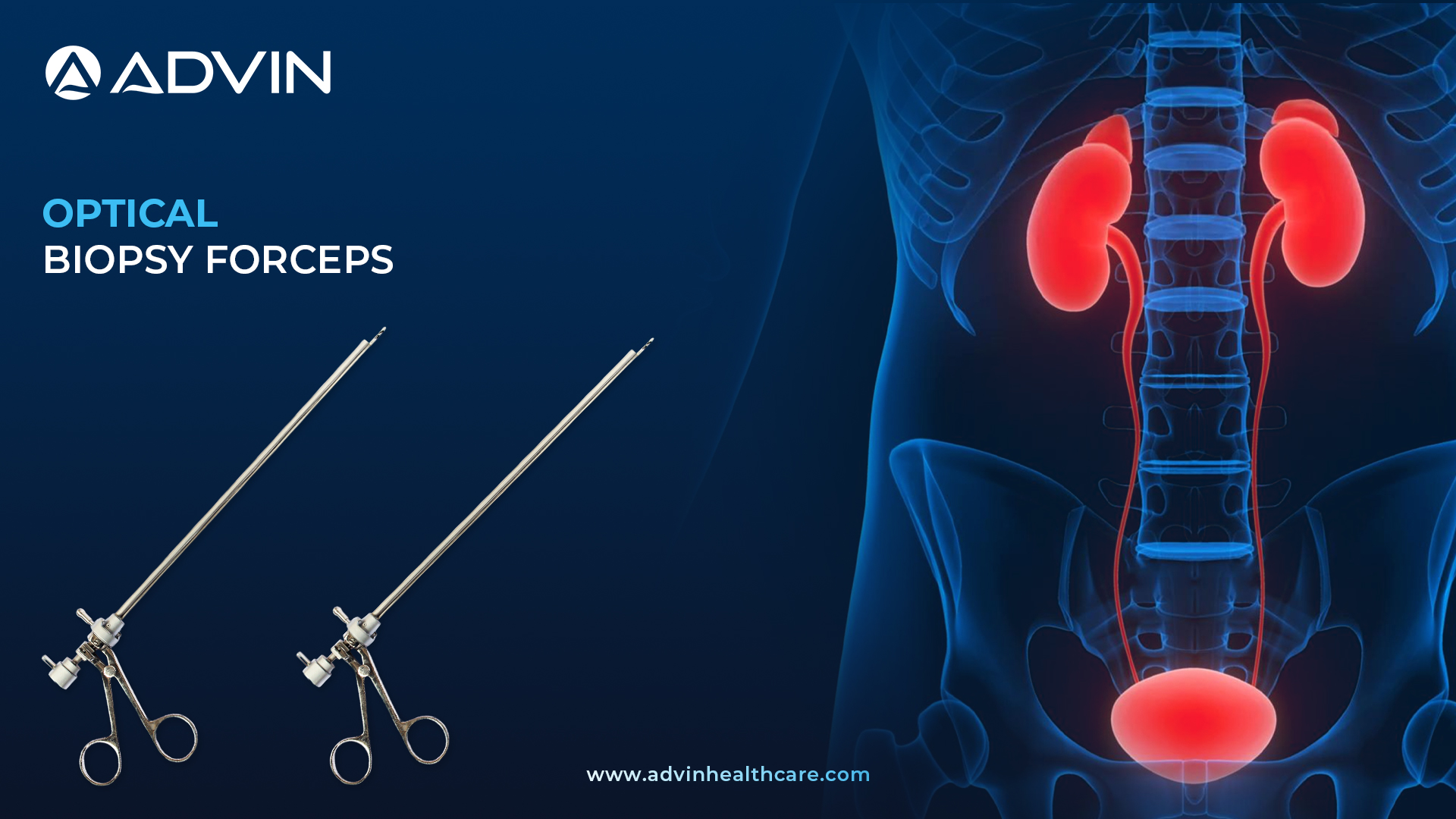
Optical Biopsy Forceps – Sharp and Controlled for Optimal Sampling
Introduction to Optical Biopsy Forceps: Visual Precision in Endoscopic Tissue Sampling:
Optical Biopsy Forceps are precision instruments used in urological endoscopy to take tissue samples from the bladder or urethra. They are compatible with endoscopes, offering clear visualization during biopsy. This tool allows accurate, safe, and controlled tissue extraction.
The Evolution of Optical Biopsy Forceps: Merging Visualization with Biopsy Accuracy:
Biopsy forceps were developed to collect tissue samples for diagnostic purposes, especially in cases of bladder tumors or urethral lesions. Early forceps lacked visual control, increasing the risk of tissue damage. The introduction of optical biopsy forceps improved accuracy by integrating a channel for telescope attachment. These forceps are now widely used in modern endoscopic urology for targeted and safe biopsies.
What Are Optical Biopsy Forceps? A Brief Overview of Function and Benefits:
The Optical Biopsy Forceps by Advin Health Care are expertly designed for precise and safe tissue sampling during endoscopic urology procedures. These forceps allow the telescope to be inserted through their shaft, providing direct vision while collecting the specimen. Advin Health Care manufactures the instrument with high-quality stainless steel, ensuring durability and repeated sterilization. The forceps offer excellent control, minimizing trauma to surrounding tissues. They are available in multiple jaw types and sizes for use with different scopes and procedures
Clinical Applications: Enhanced Tissue Retrieval in Urology and Endoscopy:
- Bladder Tumor Biopsy
- Urethral Lesion Biopsy
- Transurethral Biopsy Procedures
- Diagnostic Cystoscopy with Biopsy
- TURBT (Transurethral Resection of Bladder Tumor – for sampling)
How to Use Optical Biopsy Forceps:
Attach the telescope to the forceps using the optical channel. Insert the combined unit into the endoscope sheath. Advance to the target site under direct vision. Open the forceps jaws, position them over the tissue, and gently close to collect the sample. Withdraw and clean the forceps thoroughly after use.
Global Utilization: Top 10 Countries Advancing Diagnostics with Optical Biopsy Forceps:
- India
- China
- United States
- Germany
- Egypt
- Brazil
- Russia
- Turkey
- Mexico
- Indonesia
Advin Product Description:
- Advin Health Care is a leading manufacturer of Optical Biopsy Forceps, offering precision-crafted, vision-assisted instruments designed for efficient and accurate tissue sampling in urology procedures.
- Flexible biopsy forceps with movable cup-shaped jaws, used to obtain biopsy specimens by introduction through a specially designed endoscope.
- Flexible Biopsy Forceps are made of high quality stainless steel to provide physicians with a product of high longevity and a smooth cup operation in any situation.
- Flexible Biopsy Forceps is also called as Flexible Cystoscopy Biopsy Forceps, Reusable Flexible Biopsy forceps and Flexible Cup Biopsy Forceps.
Advanced Features:
- Superior cutting performance
- J Less bleeding
- Reusable Flexible Blopsy forceps
- Autoclavable
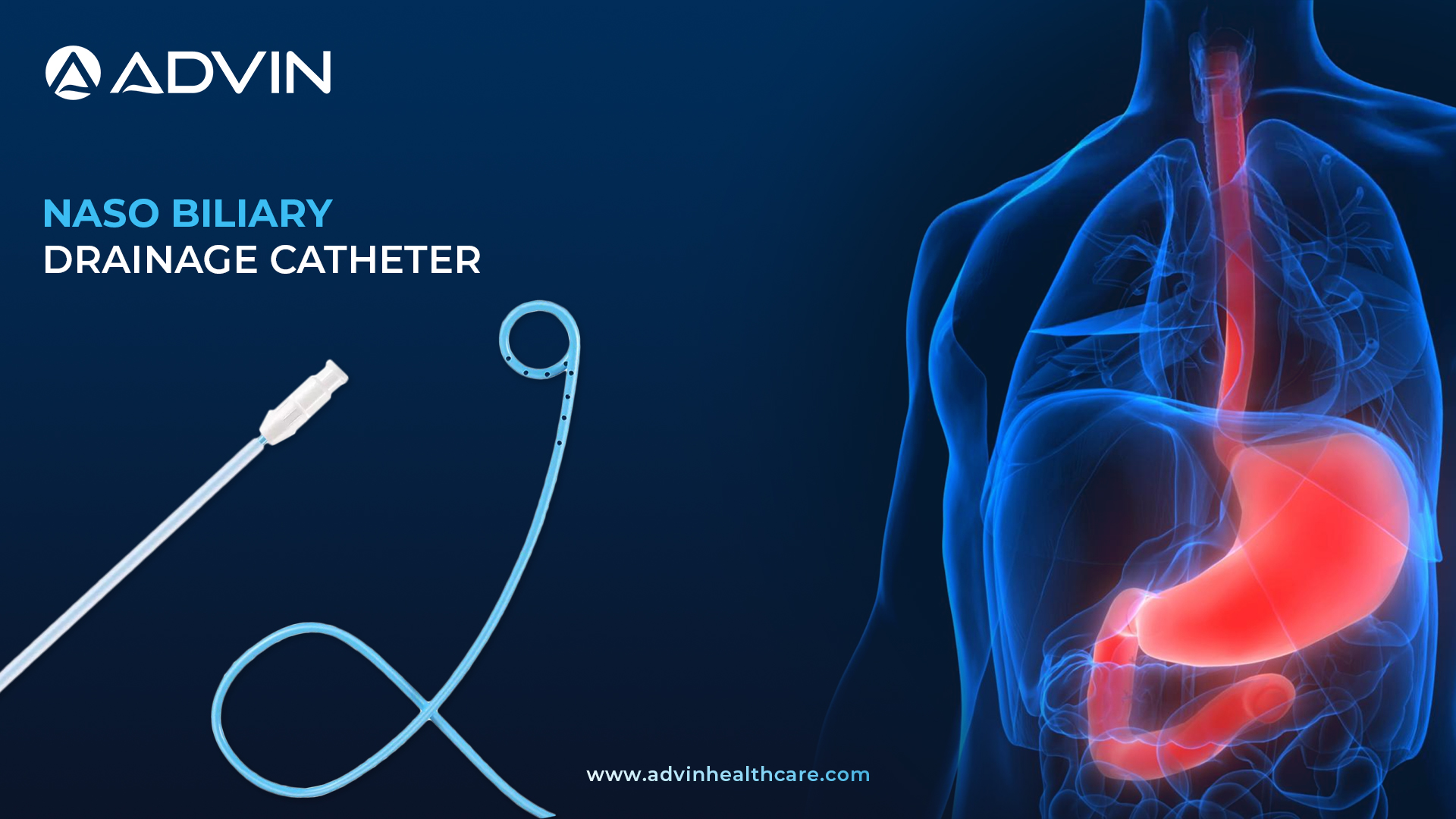
Naso Biliary Drainage Catheter - A product for Non-Surgical Biliary Relief
Understanding the Naso Biliary Drainage Catheter
A Naso Biliary Drainage Catheter is a soft, flexible tube designed to drain bile from the biliary duct system through the nose. It is commonly used in cases of obstructive jaundice and cholangitis for decompression. The catheter plays a vital role in relieving bile duct obstruction and preventing further infection.
Evolution of Naso Biliary Drainage Catheters
The concept of biliary drainage dates back to early developments in endoscopic retrograde cholangiopancreatography (ERCP). Over time, catheters evolved to offer less invasive and more comfortable options for patients. The naso biliary drainage approach became widely accepted due to its ability to provide continuous external drainage while avoiding major surgery.
Naso Biliary Drainage Catheter Description
Advin Health Care offers a high-quality Naso Biliary Drainage Catheter designed for efficient bile duct drainage. This product is widely used in the management of biliary obstructions and strictures. The catheter includes radiopaque markings for better visualization during X-ray-guided placement. Advin Health Care ensures each catheter is sterile, single-use, and individually packed to maintain hygiene standards. The pigtail tip design helps in secure placement and effective drainage.
Surgeries and Procedures Where It Is Used
- Endoscopic Retrograde Cholangiopancreatography (ERCP)
- Treatment of cholangitis
- Management of obstructive jaundice
- Post-operative bile duct stricture drainage
- Biliary decompression procedures
Instructions for Safe and Effective Use
- Connect the catheter to the drainage bag or system before insertion.
- Insert the catheter carefully through the nasal passage under fluoroscopic guidance.
- Ensure correct positioning using radiopaque markers.
- Monitor the patient and drainage flow regularly.
- Remove the catheter as per physician’s recommendation or after resolution of obstruction.
Market Demand & Global Trends
The use of Naso Biliary Drainage Catheters is rising globally due to the growing preference for minimally invasive procedures like ERCP. Hospitals and gastroenterology units now rely more on external biliary drainage for better control and patient monitoring. This trend is strong in countries such as India, China, and Brazil, where access to endoscopic care is expanding. Meanwhile, the U.S. and Germany maintain steady demand through established clinical protocols and advanced technologies. As awareness increases, the global need for effective biliary drainage solutions continues to grow.
Advin Naso Biliary Drainage Catheter Overview
Advin Health Care is a prominent manufacturer of Naso Biliary Drainage Catheters, offering innovative solutions that meet the highest medical standards. These catheters are crafted for precise bile duct drainage and patient comfort.
Advanced Features:
- Radiopaque marking for x- ray visualization
- Pigtail type
- Sterile, single use only
- Individually packed
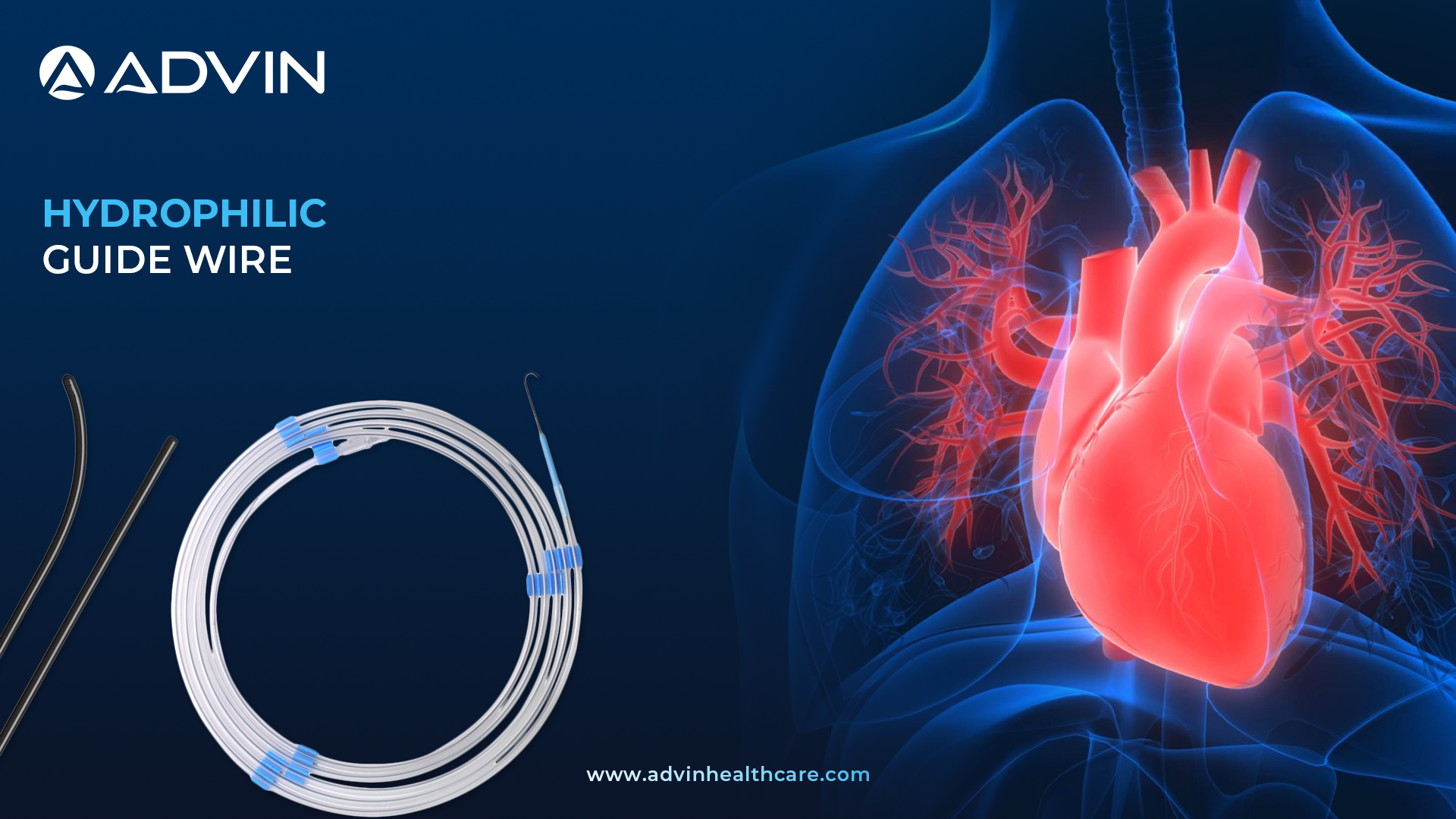
Hydrophilic Guide Wire – Trusted Solution for Interventional Cardiology
Hydrophilic Guide Wire
The Hydrophilic Guide Wire is a flexible medical device used to navigate blood vessels during cardiology procedures. It has a special coating that becomes slippery when wet, helping doctors pass it through narrow or blocked arteries smoothly. This guide wire is essential in many hospital surgeries to improve accuracy and safety.
Product History
Hydrophilic guide wires were developed to address the challenges of navigating tight or tortuous blood vessels. Before their invention, doctors faced difficulties due to friction and stiffness in traditional wires. Over time, improvements in coating materials enhanced their slipperiness and durability. Today, they are widely used in cardiology for safer and more efficient procedures.
Superior Flexibility for Enhanced Vascular Access
Advin Health Care provides high-quality Hydrophilic Guide Wires designed to ensure smooth and safe passage through blood vessels. These guide wires reduce friction significantly, which helps in minimizing vessel trauma during procedures. Advin Health Care’s product is made with a durable core and a specialized hydrophilic coating that activates when wet. This feature enhances maneuverability and precision in complex surgeries. The guide wires are suitable for various cardiology applications, providing reliable performance every time.
Surgeries Related to Product
- Coronary angioplasty
- Peripheral angioplasty
- Percutaneous coronary intervention (PCI)
- Balloon angioplasty
- Stent placement procedures
Instructions for Use
Before use, soak the Hydrophilic Guide Wire in sterile saline to activate the slippery coating. Carefully insert the wire into the blood vessel and gently guide it to the target area. Avoid applying excessive force to prevent vessel injury. After positioning, use it as a pathway to deliver other devices like catheters or balloons. Always handle with care and follow sterile procedures.
Top 10 Countries of Surgeries
- United States
- Germany
- Japan
- India
- China
- Brazil
- United Kingdom
- South Korea
- France
- Canada
Advin Product Description
Advin Health Care is a leading manufacturer of Hydrophilic Guide Wires, known for their quality and reliability. Their products help healthcare professionals perform cardiology procedures safely and effectively, ensuring better patient outcomes.
Hydrophilic Guide Wire – Key Features
- Precise tip delivery to target vessel
- Transmits rotational force to distal tip
- Supports advancement of balloon catheters/devices
- Helps locate and enter true lumen
- Distal tip retains shape memory
- Available in hydrophilic and non-coated versions
- Coil-tipped for better flexibility and control
- Hydrophilic coating enhances track ability.
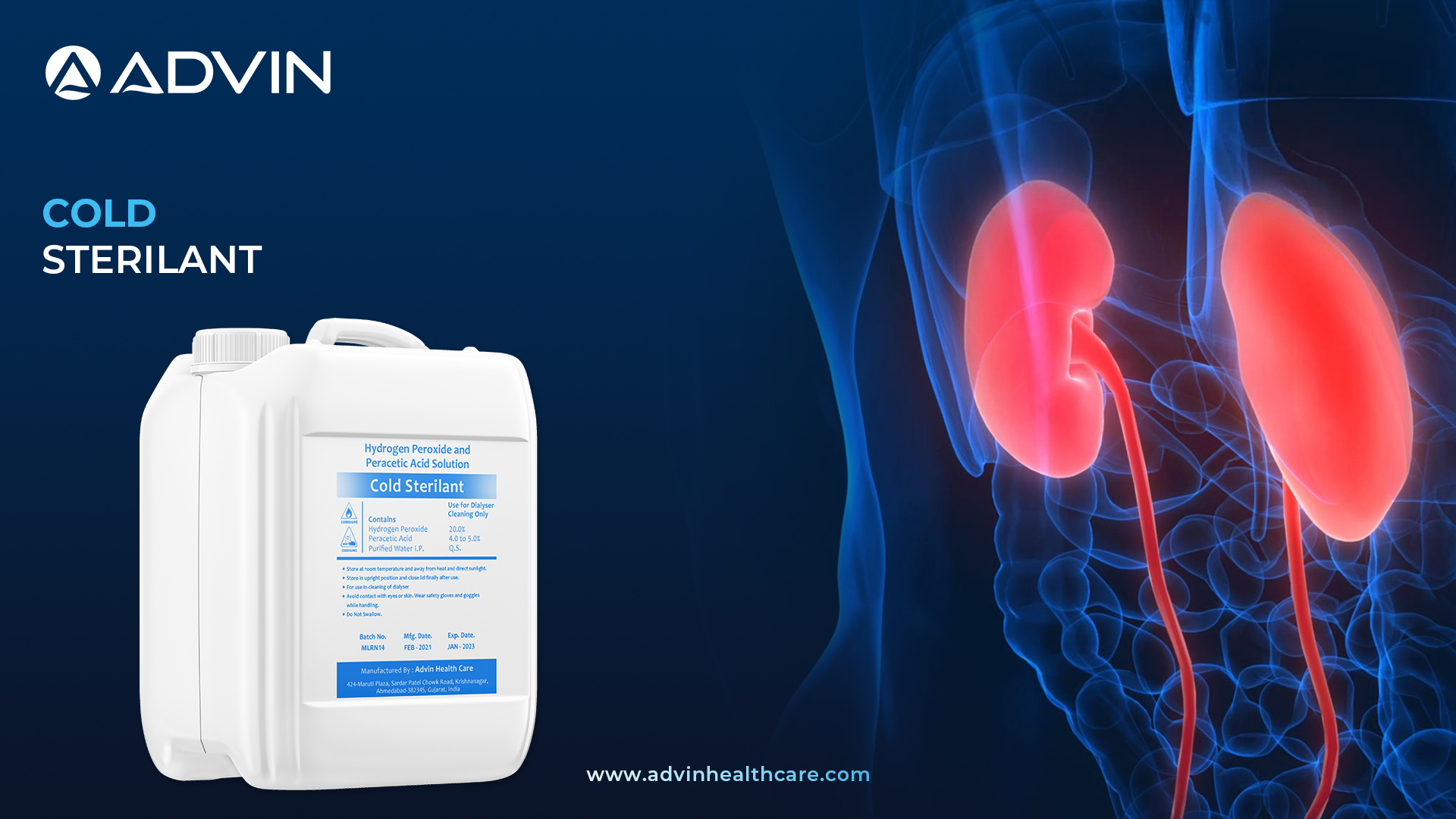
Cold Sterilant - Complete Cleaning & Disinfection in One Step
Cold Sterilant
Cold Sterilant is a chemical solution used to disinfect medical equipment without using heat. It is ideal for sterilizing heat-sensitive dialysis instruments and components. The solution helps maintain a clean and safe environment in hospitals and clinics.
Product History
Cold Sterilants have been in use since the mid-20th century when medical professionals needed an effective way to sterilize delicate instruments. Over time, formulations improved to become more efficient and less toxic. Modern Cold Sterilants are now widely used in dialysis units due to their ability to eliminate a broad range of microorganisms. These products are essential in ensuring infection control without damaging medical equipment.
Cold Sterilant – Trusted Protection for Heat-Sensitive Medical Devices
Cold Sterilant by Advin Health Care is specially formulated for sterilizing dialysis equipment that cannot be exposed to heat. It offers a broad-spectrum antimicrobial effect, ensuring the safety of both patients and healthcare providers. Advin Health Care provides this product with high-quality standards and effectiveness in mind. Its no-rinse, ready-to-use formulation saves time and maintains equipment longevity. The product is ideal for use in clinics, hospitals, and dialysis centers globally.
Surgeries Related to Product
- Hemodialysis procedures
- Peritoneal dialysis
- Kidney transplant preparation
- Catheter placement surgeries
- Vascular access surgeries (e.g., AV fistula or graft creation)
Instructions for Use
- Wear protective gloves and eyewear before handling the solution.
- Rinse all visible residues from the equipment to be sterilized.
- Fully immerse or fill the medical device with Cold Sterilant.
- Allow the equipment to soak for the recommended contact time (as per label instructions).
- After soaking, remove and allow equipment to air dry or rinse if specified.
Top 10 Countries of Surgeries
- United States
- India
- China
- Brazil
- Germany
- Japan
- Mexico
- Russia
- Turkey
- South Korea
Advin Cold Sterilant Description
- Advin Health Care is a prominent manufacturer of Cold Sterilant, offering safe and efficient sterilization solutions for dialysis and other medical equipment.
- Cold Sterilant is a validated solution specifically designed for reprocessing hollow-fiber dialyzers.
- It is a carefully balanced mixture of hydrogen peroxide, peracetic acid, and acetic acid.
- Formulated with peracetic acid as the primary active ingredient, this powerful solution ensures high-level disinfection.
- It has been widely accepted by the medical community for its effectiveness in eliminating a broad spectrum of microorganisms.
- This cold sterilant offers a reliable and safe method for sterilizing heat-sensitive dialysis equipment.
Advanced Features
- Effectively cleans hollow fibers while sterilizing and disinfecting the dialyzer
- Enhances overall cleanliness of dialysis equipment
- Provides complete and reliable disinfection
- Available in a convenient 5-liter can
- Suitable for use in hospitals and laboratories
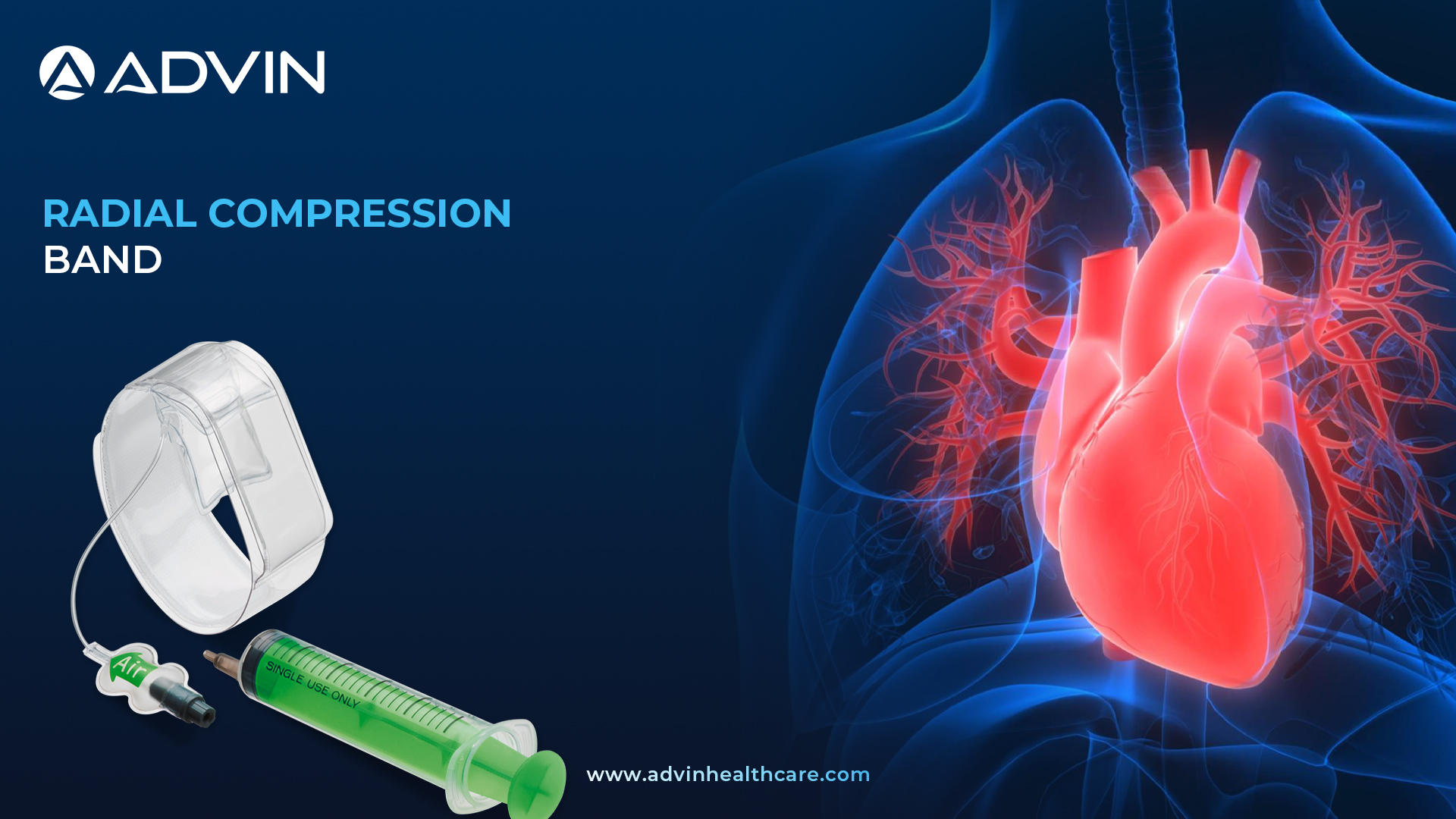
Radial Compression Band – Precision Pressure for Safe Radial Artery Management
Radial Compression Band
A Radial Compression Band is a medical device used after procedures involving the radial artery, like cardiac catheterization. It helps stop bleeding at the wrist and promotes healing. This band is commonly used in hospitals and heart care centers after minimally invasive heart procedures.
Product History
The Radial Compression Band was developed to improve patient safety and comfort after transradial access procedures. Before its introduction, manual compression was used, which was time-consuming and uncomfortable. With this device, hospitals saw fewer complications and quicker recovery times. It has now become standard practice in many heart care centers around the world.
Radial Compression Band – Controlled Compression for Reliable Hemostasis
Advin Health Care provides a high-quality Radial Compression Band that ensures safe and efficient hemostasis following radial artery access. The band is designed to maintain controlled pressure without compromising blood flow to the hand. It reduces the risk of complications such as hematoma or arterial occlusion. Advin Health Care offers this product with an easy-to-use design and secure fit for all wrist sizes. Its transparent band also allows for visual inspection without removal, ensuring better monitoring and care.
Surgeries Related to Product
- Cardiac catheterization
- Coronary angiography
- Percutaneous coronary intervention (PCI)
- Electrophysiology procedures via radial access
- Radial artery blood sampling or cannulation
Instructions for Use
- Place the Radial Compression Band around the patient’s wrist after the procedure.
- Align the compression pad directly over the puncture site.
- Inflate the band gradually to apply controlled pressure.
- Monitor the site and adjust pressure as needed to maintain hemostasis.
- Deflate and remove the band once bleeding has stopped and medical staff approves removal.
Top 10 Countries of Surgeries
- United States
- China
- India
- Germany
- Japan
- Brazil
- United Kingdom
- France
- Russia
- Italy
Advin Radial Compression Band Description
Advin Health Care is a leading manufacturer of Radial Compression Band, offering a reliable and safe solution for post-catheterization hemostasis in cardiac procedures.
Features of Radial Compression Band
- Full Transparency
- High-Adhesion Tape
- Pressure-Control Balloon
- Universal Luer Lock Compatibility